Specificity of Protein Covalent Modification by the Electrophilic Proteasome Inhibitor Carfilzomib in Human Cells
- PMID: 27503896
- PMCID: PMC5054346
- DOI: 10.1074/mcp.M116.059709
Specificity of Protein Covalent Modification by the Electrophilic Proteasome Inhibitor Carfilzomib in Human Cells
Abstract
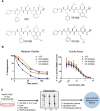
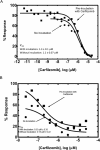
Carfilzomib (CFZ) is a second-generation proteasome inhibitor that is Food and Drug Administration and European Commission approved for the treatment of relapsed or refractory multiple myeloma. CFZ is an epoxomicin derivative with an epoxyketone electrophilic warhead that irreversibly adducts the catalytic threonine residue of the β5 subunit of the proteasome. Although CFZ produces a highly potent, sustained inactivation of the proteasome, the electrophilic nature of the drug could potentially produce off-target protein adduction. To address this possibility, we synthesized an alkynyl analog of CFZ and investigated protein adduction by this analog in HepG2 cells. Using click chemistry coupled with streptavidin based IP and shotgun tandem mass spectrometry (MS/MS), we identified two off-target proteins, cytochrome P450 27A1 (CYP27A1) and glutathione S-transferase omega 1 (GSTO1), as targets of the alkynyl CFZ probe. We confirmed the adduction of CYP27A1 and GSTO1 by streptavidin capture and immunoblotting methodology and then site-specifically mapped the adducts with targeted MS/MS methods. Although CFZ adduction of CYP27A1 and GSTO1 in vitro decreased the activities of these enzymes, the small fraction of these proteins modified by CFZ in intact cells should limit the impact of these off-target modifications. The data support the high selectivity of CFZ for covalent modification of its therapeutic targets, despite the presence of a reactive electrophile. The approach we describe offers a generalizable method to evaluate the safety profile of covalent protein-modifying therapeutics.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Conflict of interest statement
of Potential Conflicts of Interest: D.C.L. received research funding from Onyx Pharmaceuticals; E.L., J.T, and H.W. are employed by Onyx Pharmaceuticals.
Figures


Similar articles
-
Using protein turnover assay to explore the drug mechanism of Carfilzomib.Acta Biochim Biophys Sin (Shanghai). 2024 Jul 8;57(2):209-222. doi: 10.3724/abbs.2024104. Acta Biochim Biophys Sin (Shanghai). 2024. PMID: 38978505 Free PMC article.
-
A6 Peptide-Tagged Core-Disulfide-Cross-Linked Micelles for Targeted Delivery of Proteasome Inhibitor Carfilzomib to Multiple Myeloma In Vivo.Biomacromolecules. 2020 Jun 8;21(6):2049-2059. doi: 10.1021/acs.biomac.9b01790. Epub 2020 May 5. Biomacromolecules. 2020. PMID: 32338875
-
Ternary Polypeptide Nanoparticles with Improved Encapsulation, Sustained Release, and Enhanced In Vitro Efficacy of Carfilzomib.Pharm Res. 2020 Oct 6;37(11):213. doi: 10.1007/s11095-020-02922-9. Pharm Res. 2020. PMID: 33025286
-
From epoxomicin to carfilzomib: chemistry, biology, and medical outcomes.Nat Prod Rep. 2013 May;30(5):600-4. doi: 10.1039/c3np20126k. Nat Prod Rep. 2013. PMID: 23575525 Free PMC article. Review.
-
Carfilzomib: a second-generation proteasome inhibitor for the treatment of multiple myeloma.Am J Health Syst Pharm. 2015 Mar 1;72(5):353-60. doi: 10.2146/ajhp130281. Am J Health Syst Pharm. 2015. PMID: 25694410 Review.
Cited by
-
Human cytochrome P450 enzymes 5-51 as targets of drugs and natural and environmental compounds: mechanisms, induction, and inhibition - toxic effects and benefits.Drug Metab Rev. 2018 Aug;50(3):256-342. doi: 10.1080/03602532.2018.1483401. Drug Metab Rev. 2018. PMID: 30717606 Free PMC article. Review.
-
Regio- and Stereospecific Synthesis of Oridonin D-Ring Aziridinated Analogues for the Treatment of Triple-Negative Breast Cancer via Mediated Irreversible Covalent Warheads.J Med Chem. 2018 Apr 12;61(7):2737-2752. doi: 10.1021/acs.jmedchem.7b01514. Epub 2018 Mar 20. J Med Chem. 2018. PMID: 29528645 Free PMC article.
-
Development of Novel Epoxyketone-Based Proteasome Inhibitors as a Strategy To Overcome Cancer Resistance to Carfilzomib and Bortezomib.J Med Chem. 2019 May 9;62(9):4444-4455. doi: 10.1021/acs.jmedchem.8b01943. Epub 2019 Apr 19. J Med Chem. 2019. PMID: 30964987 Free PMC article.
-
Histopathological and biochemical profiling of Carfilzomib-loaded Fe-Co MOFs.Discov Nano. 2025 Aug 14;20(1):135. doi: 10.1186/s11671-025-04334-3. Discov Nano. 2025. PMID: 40804566 Free PMC article.
-
A dual inhibitor of the proteasome catalytic subunits LMP2 and Y attenuates disease progression in mouse models of Alzheimer's disease.Sci Rep. 2019 Dec 5;9(1):18393. doi: 10.1038/s41598-019-54846-z. Sci Rep. 2019. PMID: 31804556 Free PMC article.
References
-
- Noe M. C., and Gilbert A. M. (2012) Targeted covalent enzyme inhibitors. Ann. Reports Med. Chem. 47, 413–439
-
- Singh J., Petter R. C., Baillie T. A., and Whitty A. (2011) The resurgence of covalent drugs. Nat. Rev. Drug Discov. 10, 307–317 - PubMed
-
- Mah R., Thomas J. R., and Shafer C. M. (2014) Drug discovery considerations in the development of covalent inhibitors. Bioorg. Med. Chem. Lett. 24, 33–39 - PubMed
-
- Gayko U., Fung M., Clow F., Sun S., Faust E., Price S., James D., Doyle M., Bari S., and Zhuang S. H. (2015) Development of the Bruton's tyrosine kinase inhibitor ibrutinib for B cell malignancies. Ann. N.Y. Acad. Sci. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

