Age-Related Decrease in Heat Shock 70-kDa Protein 8 in Cerebrospinal Fluid Is Associated with Increased Oxidative Stress
- PMID: 27507943
- PMCID: PMC4960228
- DOI: 10.3389/fnagi.2016.00178
Age-Related Decrease in Heat Shock 70-kDa Protein 8 in Cerebrospinal Fluid Is Associated with Increased Oxidative Stress
Abstract
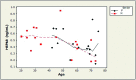
Age-associated declines in protein homeostasis mechanisms ("proteostasis") are thought to contribute to age-related neurodegenerative disorders. The increased oxidative stress which occurs with aging can activate a key proteostatic process, chaperone-mediated autophagy. This study investigated age-related alteration in cerebrospinal fluid (CSF) concentrations of heat shock 70-kDa protein 8 (HSPA8), a molecular chaperone involved in proteostatic mechanisms including chaperone-mediated autophagy, and its associations with indicators of oxidative stress (8-hydroxy-2'-deoxyguanosine [8-OHdG] and 8-isoprostane) and total anti-oxidant capacity. We examined correlations between age, HSPA8, 8-OHdG, 8-isoprostane, and total antioxidant capacity (TAC) in CSF samples from 34 healthy subjects ranging from 20 to 75 years of age. Age was negatively associated with HSPA8 (ρ = -0.47; p = 0.005). An age-related increase in oxidative stress was indicated by a positive association between age and 8-OHdG (ρ = 0.61; p = 0.0001). HSPA8 was moderately negatively associated with 8-OHdG (ρ = -0.58; p = 0.0004). Age and HSPA8 were weakly associated with 8-isoprostane and TAC (range of ρ values: -0.15 to 0.16). Our findings in this exploratory study suggest that during healthy aging, CSF HSPA8 may decrease, perhaps due in part to an increase in oxidative stress. Our results also suggest that 8-OHdG may be more sensitive than 8-isoprostane for measuring oxidative stress in CSF. Further studies are indicated to determine if our findings can be replicated with a larger cohort, and if the age-related decrease in HSPA8 in CSF is reflected by a similar change in the brain.
Keywords: 8-hydroxydeoxyguanosine; 8-isoprostane; HSPA8; aging; cerebrospinal fluid; oxidative stress.
Figures

Similar articles
-
Cerebrospinal Fluid Concentration of Key Autophagy Protein Lamp2 Changes Little During Normal Aging.Front Aging Neurosci. 2018 May 8;10:130. doi: 10.3389/fnagi.2018.00130. eCollection 2018. Front Aging Neurosci. 2018. PMID: 29867441 Free PMC article.
-
Increased Oxidative Stress Markers in Cerebrospinal Fluid from Healthy Subjects with Parkinson's Disease-Associated LRRK2 Gene Mutations.Front Aging Neurosci. 2017 Apr 3;9:89. doi: 10.3389/fnagi.2017.00089. eCollection 2017. Front Aging Neurosci. 2017. PMID: 28420983 Free PMC article.
-
The evaluation of oxidative DNA damage in children with brain damage using 8-hydroxydeoxyguanosine levels.Brain Dev. 2008 Feb;30(2):131-6. doi: 10.1016/j.braindev.2007.07.005. Epub 2007 Sep 4. Brain Dev. 2008. PMID: 17766071
-
Influence of Normal Aging on Brain Autophagy: A Complex Scenario.Front Aging Neurosci. 2019 Mar 11;11:49. doi: 10.3389/fnagi.2019.00049. eCollection 2019. Front Aging Neurosci. 2019. PMID: 30914945 Free PMC article. Review.
-
Redox regulation of heat shock protein expression in aging and neurodegenerative disorders associated with oxidative stress: a nutritional approach.Amino Acids. 2003 Dec;25(3-4):437-44. doi: 10.1007/s00726-003-0048-2. Epub 2003 Nov 7. Amino Acids. 2003. PMID: 14661103 Review.
Cited by
-
Identification and validation of autophagy-related genes in primary open-angle glaucoma.BMC Med Genomics. 2023 Nov 15;16(1):287. doi: 10.1186/s12920-023-01722-5. BMC Med Genomics. 2023. PMID: 37968618 Free PMC article.
-
Age-related neurodegenerative diseases.J Cell Physiol. 2020 Apr;235(4):3131-3141. doi: 10.1002/jcp.29248. Epub 2019 Sep 25. J Cell Physiol. 2020. PMID: 31556109 Free PMC article. Review.
-
Gene Expression Profiles of Human Cerebral Organoids Identify PPAR Pathway and PKM2 as Key Markers for Oxygen-Glucose Deprivation and Reoxygenation.Front Cell Neurosci. 2021 Jun 8;15:605030. doi: 10.3389/fncel.2021.605030. eCollection 2021. Front Cell Neurosci. 2021. PMID: 34168538 Free PMC article.
-
Impact of Chaperone-Mediated Autophagy in Brain Aging: Neurodegenerative Diseases and Glioblastoma.Front Aging Neurosci. 2021 Jan 28;12:630743. doi: 10.3389/fnagi.2020.630743. eCollection 2020. Front Aging Neurosci. 2021. PMID: 33633561 Free PMC article.
-
SARS-CoV-2 infection increases the gene expression profile for Alzheimer's disease risk.Mol Ther Methods Clin Dev. 2022 Dec 8;27:217-229. doi: 10.1016/j.omtm.2022.09.007. Epub 2022 Sep 24. Mol Ther Methods Clin Dev. 2022. PMID: 36187720 Free PMC article.
References
-
- Calabrese V., Scapagnini G., Ravagna A., Colombrita C., Spadaro F., Butterfield D. A., et al. (2004). Increased expression of heat shock proteins in rat brain during aging: relationship with mitochondrial function and glutathione redox state. Mech. Ageing Dev. 125 325–335. 10.1016/j.mad.2004.01.003 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

