Clinical Effects and Safety of Direct-Acting Insulin Analogs in Patients with Type 1 Diabetes: A Nation-Wide Observational Cohort Study
- PMID: 27517679
- PMCID: PMC5014797
- DOI: 10.1007/s13300-016-0191-x
Clinical Effects and Safety of Direct-Acting Insulin Analogs in Patients with Type 1 Diabetes: A Nation-Wide Observational Cohort Study
Abstract
Introduction: Studies comparing direct-acting insulin analogs (DAIs) in terms of effectiveness and long-term safety are scarce. Our aim was to explore these variables in clinical practice among patients with type 1 diabetes, including the elderly and those with renal impairment.
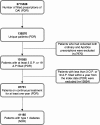
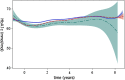
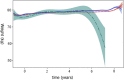
Methods: We linked four national registers in a population-based cohort study. Patients with type 1 diabetes and continuous use of all currently available DAIs (lispro, aspart, or glulisine) in 2005-2013 were monitored for up to 7.5 years. Inverse probability of treatment weighting was used to adjust for differences in baseline characteristics between treatment groups. Unadjusted mean HbA1c and weights were plotted. Hazard ratios and 95% confidence intervals of cardiovascular events (CVEs) and mortality were estimated using Cox proportional hazards regression models.
Results: We included 41,165 patients-14,047 lispro, 26,813 aspart, and 305 glulisine users. At baseline, the mean age was highest among glulisine users (49.4 years), followed by 41.0 years for lispro users and 40.1 years for aspart users. A total of 9.2% of the patients were 65 years or older. Diabetes duration was shortest among glulisine users (11.6 years), followed by 15.4 years for aspart users and 19.5 years for lispro users. The mean HbA1c and weights during the follow-up period were similar. The numerical differences at baseline were subsequently adjusted for. There were no significant differences between groups regarding hyperglycemia requiring hospitalization, CVE, or mortality, while Cox regression suggested lower rates of hypoglycemia among glulisine users. Severe hypoglycemia was more common, and severe hyperglycemia was less common among patients aged 65 years or older, while severe hypoglycemia and hyperglycemia were more common in patients with low renal function (estimated glomerular filtration rate).
Conclusion: There were no pronounced differences in effectiveness and long-term cardiovascular safety and mortality between the DAIs, although there were some differences in clinical characteristics between patients using the three types of insulin. Severe hypoglycemia was more common among older patients, while severe hypoglycemia and hyperglycemia were more common among patients with impaired renal function.
Funding: Sanofi.
Keywords: Cohort study; Effectiveness; Elderly; Insulin analogs; Insulin aspart; Insulin glulisine; Insulin lispro; Renal failure; Safety; Type 1 diabetes.
Figures
Similar articles
-
Clinical effects, cardiovascular and renal outcomes associated with rapid-acting insulin analogs among individuals with type 2 diabetes: a nation-wide observational cohort study.Clin Diabetes Endocrinol. 2017 Jun 19;3:5. doi: 10.1186/s40842-017-0043-2. eCollection 2017. Clin Diabetes Endocrinol. 2017. PMID: 28702259 Free PMC article.
-
A comparison of the rapid-acting insulin analogue glulisine with lispro and aspart for the pump treatment of patients with type 1 diabetes.Acta Diabetol. 2022 Nov;59(11):1453-1460. doi: 10.1007/s00592-022-01939-3. Epub 2022 Aug 7. Acta Diabetol. 2022. PMID: 35933650
-
Comparison of pharmacokinetic properties, physicochemical stability, and pump compatibility of 3 rapid-acting insulin analogues-aspart, lispro, and glulisine.Endocr Pract. 2011 Mar-Apr;17(2):271-80. doi: 10.4158/EP10260.RA. Endocr Pract. 2011. PMID: 21134878 Review.
-
Biphasic insulin aspart 30: literature review of adverse events associated with treatment.Clin Ther. 2005;27 Suppl B:S75-88. doi: 10.1016/j.clinthera.2005.11.022. Clin Ther. 2005. PMID: 16519039 Review.
-
Insulin glulisine: an evaluation of its pharmacodynamic properties and clinical application.Ann Pharmacother. 2009 Apr;43(4):658-68. doi: 10.1345/aph.1E662. Epub 2009 Mar 31. Ann Pharmacother. 2009. PMID: 19336657 Review.
Cited by
-
Clinical effects, cardiovascular and renal outcomes associated with rapid-acting insulin analogs among individuals with type 2 diabetes: a nation-wide observational cohort study.Clin Diabetes Endocrinol. 2017 Jun 19;3:5. doi: 10.1186/s40842-017-0043-2. eCollection 2017. Clin Diabetes Endocrinol. 2017. PMID: 28702259 Free PMC article.
-
Investigation of Severe Hypoglycemia Risk Among Patients with Diabetes Treated with Ultra-Rapid Lispro in Japan.Adv Ther. 2025 Jan;42(1):413-426. doi: 10.1007/s12325-024-03050-1. Epub 2024 Nov 21. Adv Ther. 2025. PMID: 39570544 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

