Suppression of the inflammatory response by disease-inducible interleukin-10 gene therapy in a three-dimensional micromass model of the human synovial membrane
- PMID: 27519904
- PMCID: PMC4983024
- DOI: 10.1186/s13075-016-1083-1
Suppression of the inflammatory response by disease-inducible interleukin-10 gene therapy in a three-dimensional micromass model of the human synovial membrane
Abstract
Background: Gene therapy has the potential to provide long-term production of therapeutic proteins in the joints of osteoarthritis (OA) patients. The objective of this study was to analyse the therapeutic potential of disease-inducible expression of anti-inflammatory interleukin-10 (IL-10) in the three-dimensional micromass model of the human synovial membrane.
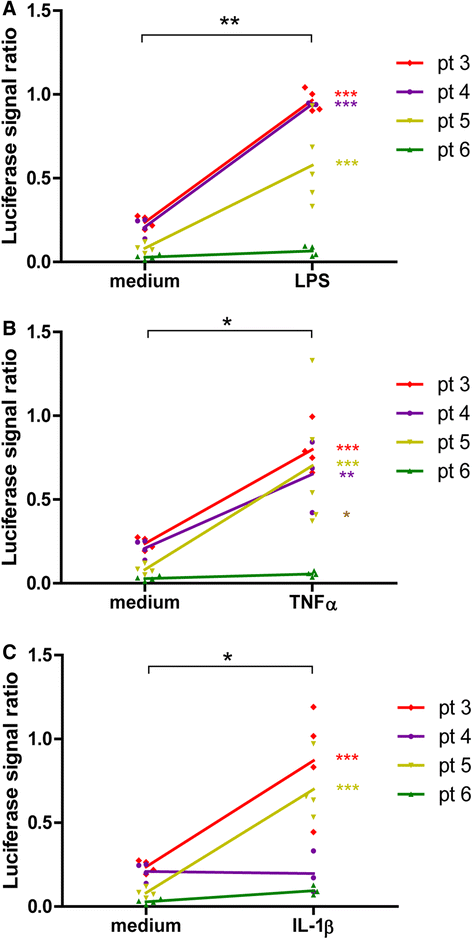
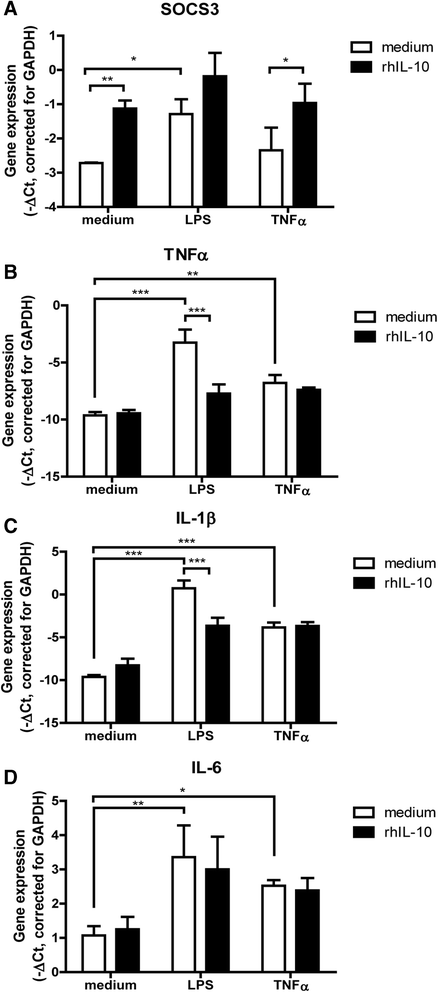
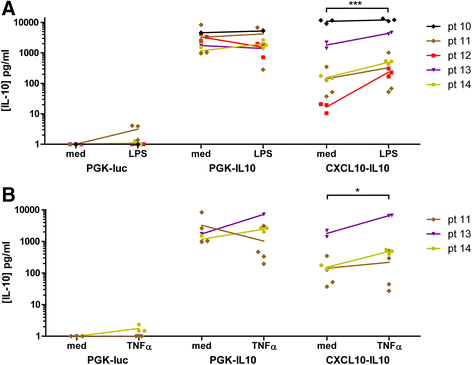
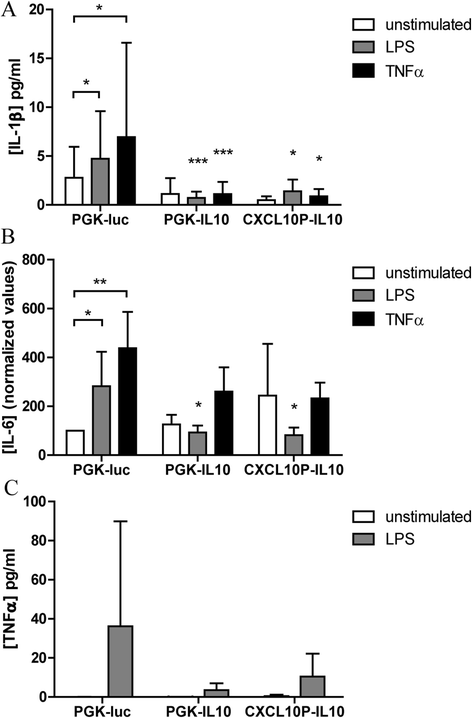
Methods: Synovial tissue samples from OA patients were digested and the cells were mixed with Matrigel to obtain 3D micromasses. The CXCL10 promoter combined with the firefly luciferase reporter in a lentiviral vector was used to determine the response of the CXCL10 promoter to tumour necrosis factor alpha (TNF-α), interleukin-1β (IL-1β) and lipopolysaccharide (LPS). The effects of recombinant IL-10 on gene expression were determined by quantitative PCR. The production of IL-10 from the CXCL10p-IL10 vector and the effects on pro-inflammatory cytokine production were assessed by multiplex ELISA.
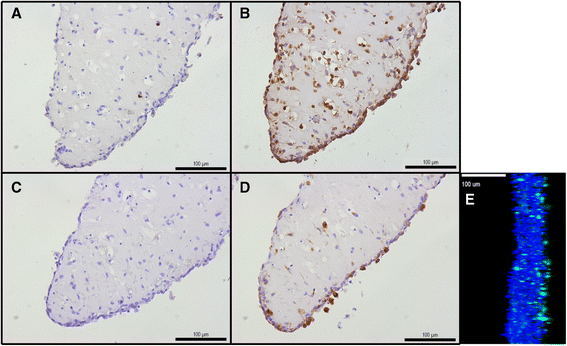
Results: Micromasses made from whole synovial membrane cell suspensions form a distinct surface composition containing macrophage and fibroblast-like synoviocytes thus mimicking the synovial lining. This lining can be transduced by lentiviruses and allow CXCL-10 promoter-regulated transgene expression. Adequate amounts of IL-10 transgene were produced after stimulation with pro-inflammatory factors able to reduce the production of synovial IL-1β and IL-6.
Conclusions: Synovial micromasses are a suitable model to test disease-regulated gene therapy approaches and the CXCL10p-IL10 vector might be a good candidate to decrease the inflammatory response implicated in the pathogenesis of OA.
Keywords: Cytokines; Gene therapy; Inflammation; Micromasses; Osteoarthritis; Synovium.
Figures
Similar articles
-
Disease-Regulated Gene Therapy with Anti-Inflammatory Interleukin-10 Under the Control of the CXCL10 Promoter for the Treatment of Rheumatoid Arthritis.Hum Gene Ther. 2016 Mar;27(3):244-54. doi: 10.1089/hum.2015.127. Epub 2016 Feb 16. Hum Gene Ther. 2016. PMID: 26711533
-
The role of synovial macrophages and macrophage-produced cytokines in driving aggrecanases, matrix metalloproteinases, and other destructive and inflammatory responses in osteoarthritis.Arthritis Res Ther. 2006;8(6):R187. doi: 10.1186/ar2099. Arthritis Res Ther. 2006. PMID: 17177994 Free PMC article.
-
Nerve growth factor regulation and production by macrophages in osteoarthritic synovium.Clin Exp Immunol. 2017 Nov;190(2):235-243. doi: 10.1111/cei.13007. Epub 2017 Jul 27. Clin Exp Immunol. 2017. PMID: 28677145 Free PMC article.
-
The role of cytokines in osteoarthritis pathophysiology.Biorheology. 2002;39(1-2):237-46. Biorheology. 2002. PMID: 12082286 Review.
-
The interleukin 1beta pathway in the pathogenesis of osteoarthritis.J Rheumatol. 2008 Dec;35(12):2306-12. doi: 10.3899/jrheum.080346. J Rheumatol. 2008. PMID: 18925684 Review.
Cited by
-
Protective Role of the MER Tyrosine Kinase via Efferocytosis in Rheumatoid Arthritis Models.Front Immunol. 2018 Apr 13;9:742. doi: 10.3389/fimmu.2018.00742. eCollection 2018. Front Immunol. 2018. PMID: 29706963 Free PMC article.
-
Encapsulated Mesenchymal Stromal Cell Microbeads Promote Endogenous Regeneration of Osteoarthritic Cartilage Ex Vivo.Adv Healthc Mater. 2021 Apr;10(8):e2002118. doi: 10.1002/adhm.202002118. Epub 2021 Jan 12. Adv Healthc Mater. 2021. PMID: 33434393 Free PMC article.
-
Association of Serum Vitamin D with Serum Cytokine Profile in Patients with Knee Osteoarthritis.Cartilage. 2021 Dec;13(1_suppl):1610S-1618S. doi: 10.1177/19476035211010309. Epub 2021 Apr 23. Cartilage. 2021. PMID: 33890506 Free PMC article.
-
Candidates for Intra-Articular Administration Therapeutics and Therapies of Osteoarthritis.Int J Mol Sci. 2021 Mar 30;22(7):3594. doi: 10.3390/ijms22073594. Int J Mol Sci. 2021. PMID: 33808364 Free PMC article. Review.
-
Repurposed and investigational disease-modifying drugs in osteoarthritis (DMOADs).Ther Adv Musculoskelet Dis. 2022 May 20;14:1759720X221090297. doi: 10.1177/1759720X221090297. eCollection 2022. Ther Adv Musculoskelet Dis. 2022. PMID: 35619876 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

