Developmental changes in plasticity, synaptic, glia and connectivity protein levels in rat dorsal hippocampus
- PMID: 27523749
- PMCID: PMC5050161
- DOI: 10.1016/j.nlm.2016.08.005
Developmental changes in plasticity, synaptic, glia and connectivity protein levels in rat dorsal hippocampus
Abstract
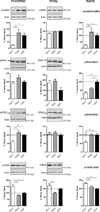
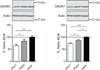
Thus far the identification and functional characterization of the molecular mechanisms underlying synaptic plasticity, learning, and memory have not been particularly dissociated from the contribution of developmental changes. Brain plasticity mechanisms have been largely identified and studied using in vitro systems mainly derived from early developmental ages, yet they are considered to be general plasticity mechanisms underlying functions -such as long-term memory- that occurs in the adult brain. Although it is possible that part of the plasticity mechanisms recruited during development is then re-recruited in plasticity responses in adulthood, systematic investigations about whether and how activity-dependent molecular responses differ over development are sparse. Notably, hippocampal-dependent memories are expressed relatively late in development, and the hippocampus undergoes and extended developmental post-natal structural and functional maturation, suggesting that the molecular mechanisms underlying hippocampal neuroplasticity may actually significantly change over development. Here we quantified the relative basal expression levels of sets of plasticity, synaptic, glia and connectivity proteins in rat dorsal hippocampus, a region that is critical for the formation of long-term explicit memories, at two developmental ages, postnatal day 17 (PN17) and PN24, which correspond to a period of relative functional immaturity and maturity, respectively, and compared them to adult age. We found that the levels of numerous proteins and/or their phosphorylation, known to be critical for synaptic plasticity underlying memory formation, including immediate early genes (IEGs), kinases, transcription factors and AMPA receptor subunits, peak at PN17 when the hippocampus is not yet able to express long-term memory. It remains to be established if these changes result from developmental basal activity or infantile learning. Conversely, among all markers investigated, the phosphorylation of calcium calmodulin kinase II α (CamKII α and of extracellular signal-regulated kinases 2 (ERK-2), and the levels of GluA1 and GluA2 significantly increase from PN17 to PN24 and then remain similar in adulthood, thus representing correlates paralleling long-term memory expression ability.
Keywords: Connectivity; Development; Hippocampus; Memory; Myelin; Plasticity; Protein; Synapse.
Copyright © 2016 Elsevier Inc. All rights reserved.
Figures

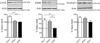


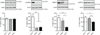

Similar articles
-
Developmental changes in plasticity, synaptic, glia, and connectivity protein levels in rat basolateral amygdala.Learn Mem. 2019 Oct 15;26(11):436-448. doi: 10.1101/lm.049866.119. Print 2019 Nov. Learn Mem. 2019. PMID: 31615855 Free PMC article.
-
Developmental changes in plasticity, synaptic, glia, and connectivity protein levels in rat medial prefrontal cortex.Learn Mem. 2018 Sep 17;25(10):533-543. doi: 10.1101/lm.047753.118. Print 2018 Oct. Learn Mem. 2018. PMID: 30224556 Free PMC article.
-
Disruption of Coordinated Presynaptic and Postsynaptic Maturation Underlies the Defects in Hippocampal Synapse Stability and Plasticity in Abl2/Arg-Deficient Mice.J Neurosci. 2016 Jun 22;36(25):6778-91. doi: 10.1523/JNEUROSCI.4092-15.2016. J Neurosci. 2016. PMID: 27335408 Free PMC article.
-
A fresh look at the role of CaMKII in hippocampal synaptic plasticity and memory.Bioessays. 2002 Mar;24(3):223-33. doi: 10.1002/bies.10057. Bioessays. 2002. PMID: 11891759 Review.
-
LTD, LTP, and the sliding threshold for long-term synaptic plasticity.Hippocampus. 1996;6(1):35-42. doi: 10.1002/(SICI)1098-1063(1996)6:1<35::AID-HIPO7>3.0.CO;2-6. Hippocampus. 1996. PMID: 8878740 Review.
Cited by
-
Obesity Control and Supplementary Nutraceuticals as Cofactors of Brain Plasticity in Multiple Sclerosis Populations.Int J Mol Sci. 2024 Oct 10;25(20):10909. doi: 10.3390/ijms252010909. Int J Mol Sci. 2024. PMID: 39456690 Free PMC article. Review.
-
Hippocampal parvalbumin interneurons play a critical role in memory development.Cell Rep. 2022 Nov 15;41(7):111643. doi: 10.1016/j.celrep.2022.111643. Cell Rep. 2022. PMID: 36384113 Free PMC article.
-
The distribution of serotonergic nerve on the hippocampus of the fruit bats (Rousettus amplexicaudatus).Vet World. 2019 Sep;12(9):1460-1466. doi: 10.14202/vetworld.2019.1460-1466. Vet World. 2019. PMID: 31749582 Free PMC article.
-
Developmental changes in plasticity, synaptic, glia, and connectivity protein levels in rat basolateral amygdala.Learn Mem. 2019 Oct 15;26(11):436-448. doi: 10.1101/lm.049866.119. Print 2019 Nov. Learn Mem. 2019. PMID: 31615855 Free PMC article.
-
Mechanisms of critical period in the hippocampus underlie object location learning and memory in infant rats.Learn Mem. 2018 Mar 15;25(4):176-182. doi: 10.1101/lm.046946.117. Print 2018 Apr. Learn Mem. 2018. PMID: 29545389 Free PMC article.
References
-
- Aigner L, Arber S, Kapfhammer JP, Laux T, Schneider C, Botteri F, Brenner HR, Caroni P. Overexpression of the neural growth-associated protein GAP-43 induces nerve sprouting in the adult nervous system of transgenic mice. Cell. 1995;83(2):269–278. - PubMed
-
- Alberini CM. Transcription factors in long-term memory and synaptic plasticity. Physiological Reviews. 2009;89(1):121–145. http://doi.org/10.1152/physrev.00017.2008. - DOI - PMC - PubMed
-
- Bähler M, Benfenati F, Valtorta F, Greengard P. The synapsins and the regulation of synaptic function. BioEssays: News and Reviews in Molecular, Cellular and Developmental Biology. 1990;12(6):259–263. http://doi.org/10.1002/bies.950120603. - DOI - PubMed
-
- Bailey CH, Kandel ER, Harris KM. Structural Components of Synaptic Plasticity and Memory Consolidation. Cold Spring Harbor Perspectives in Biology. 2015;7(7):a021758. http://doi.org/10.1101/cshperspect.a021758. - DOI - PMC - PubMed
-
- Barco A, Pittenger C, Kandel ER. CREB, memory enhancement and the treatment of memory disorders: promises, pitfalls and prospects. Expert Opin Ther Targets. 2003;7(1):101–114. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

