Kampo Medicine: Evaluation of the Pharmacological Activity of 121 Herbal Drugs on GABAA and 5-HT3A Receptors
- PMID: 27524967
- PMCID: PMC4965468
- DOI: 10.3389/fphar.2016.00219
Kampo Medicine: Evaluation of the Pharmacological Activity of 121 Herbal Drugs on GABAA and 5-HT3A Receptors
Abstract
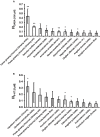
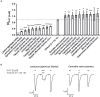
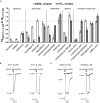
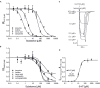
Kampo medicine is a form of Japanese phytotherapy originating from traditional Chinese medicine (TCM). During the last several decades, much attention has been paid to the pharmacological effects of these medical plants and their constituents. However, in many cases, a systematic screening of Kampo remedies to determine pharmacologically relevant targets is still lacking. In this study, a broad screening of Kampo remedies was performed to look for pharmacologically relevant 5-HT3A and GABAA receptor ligands. Several of the Kampo remedies are currently used for symptoms such as nausea, emesis, gastrointestinal motility disorders, anxiety, restlessness, or insomnia. Therefore, the pharmacological effects of 121 herbal drugs from Kampo medicine were analyzed as ethanol tinctures on heterologously expressed 5-HT3A and GABAA receptors, due to the involvement of these receptors in such pathophysiological processes. The tinctures of Lindera aggregata (radix) and Leonurus japonicus (herba) were the most effective inhibitory compounds on the 5-HT3A receptor. Further investigation of known ingredients in these compounds led to the identification of leonurine from Leonurus as a new natural 5-HT3A receptor antagonist. Several potentiating herbs (e.g., Magnolia officinalis (cortex), Syzygium aromaticum (flos), and Panax ginseng (radix)) were also identified for the GABAA receptor, which are all traditionally used for their sedative or anxiolytic effects. A variety of tinctures with antagonistic effects Salvia miltiorrhiza (radix) were also detected. Therefore, this study reveals new insights into the pharmacological action of a broad spectrum of herbal drugs from Kampo, allowing for a better understanding of their physiological effects and clinical applications.
Keywords: 5-HT3A receptor; GABAA receptor; Kampo; Leonurus japonicus (herba); Panax ginseng (radix); Salvia miltiorrhiza (radix); andrographolide; leonurine.
Figures


Similar articles
-
Identification of Glycyrrhiza as the rikkunshito constituent with the highest antagonistic potential on heterologously expressed 5-HT3A receptors due to the action of flavonoids.Front Pharmacol. 2015 Jul 3;6:130. doi: 10.3389/fphar.2015.00130. eCollection 2015. Front Pharmacol. 2015. PMID: 26191003 Free PMC article.
-
Interactions between crude drug extracts used in Japanese traditional Kampo medicines and organic anion-transporting polypeptide 2B1.J Ethnopharmacol. 2018 Mar 25;214:153-159. doi: 10.1016/j.jep.2017.12.016. Epub 2017 Dec 14. J Ethnopharmacol. 2018. PMID: 29248449
-
Pharmacological characteristics of Kampo medicine as a mixture of constituents and ingredients.J Integr Med. 2013 Jan;11(1):11-6. doi: 10.3736/jintegrmed2013003. J Integr Med. 2013. PMID: 23464641 Review.
-
Herbal medicine and false-positive results on lymphocyte transformation test.Yakugaku Zasshi. 2002 Jun;122(6):399-402. doi: 10.1248/yakushi.122.399. Yakugaku Zasshi. 2002. PMID: 12087777 Clinical Trial.
-
A review of traditional Japanese medicines and their potential mechanism of action.Curr Pharm Des. 2012;18(31):4839-53. doi: 10.2174/138161212803216924. Curr Pharm Des. 2012. PMID: 22632864 Review.
Cited by
-
Anti-Trypanosoma cruzi activity of Coptis rhizome extract and its constituents.Trop Med Health. 2023 Mar 1;51(1):12. doi: 10.1186/s41182-023-00502-2. Trop Med Health. 2023. PMID: 36859380 Free PMC article.
-
Anti-epileptic Kunitz-like peptides discovered in the branching coral Acropora digitifera through transcriptomic analysis.Arch Toxicol. 2022 Sep;96(9):2589-2608. doi: 10.1007/s00204-022-03311-4. Epub 2022 May 23. Arch Toxicol. 2022. PMID: 35604417
-
Effects of cannabinoids on ligand-gated ion channels.Front Physiol. 2022 Oct 19;13:1041833. doi: 10.3389/fphys.2022.1041833. eCollection 2022. Front Physiol. 2022. PMID: 36338493 Free PMC article. Review.
-
The Influence of Palmatine Isolated from Berberis sibiricaRadix on Pentylenetetrazole-Induced Seizures in Zebrafish.Cells. 2020 May 16;9(5):1233. doi: 10.3390/cells9051233. Cells. 2020. PMID: 32429356 Free PMC article.
-
Anti-malarial activity of traditional Kampo medicine Coptis rhizome extract and its major active compounds.Malar J. 2020 Jun 8;19(1):204. doi: 10.1186/s12936-020-03273-x. Malar J. 2020. PMID: 32513250 Free PMC article.
References
-
- Atawodi S. E., Atawodi J. C., Pfundstein B., Spiegelhalder B., Bartsch H., Owen R. (2011). Assessment of the polyphenol components and in vitro antioxidant properties of Syzygium aromaticum (L.) Merr. & Perry. Electron. J. Environ. Agric. Food Chem. 10 1970–1978.
LinkOut - more resources
Full Text Sources
Other Literature Sources

