ISL1 cardiovascular progenitor cells for cardiac repair after myocardial infarction
- PMID: 27525311
- PMCID: PMC4982472
- DOI: 10.1172/jci.insight.80920
ISL1 cardiovascular progenitor cells for cardiac repair after myocardial infarction
Abstract
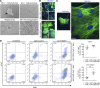
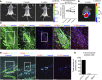
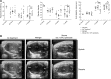
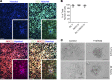
Cardiovascular progenitor cells (CPCs) expressing the ISL1-LIM-homeodomain transcription factor contribute developmentally to cardiomyocytes in all 4 chambers of the heart. Here, we show that ISL1-CPCs can be applied to myocardial regeneration following injury. We used a rapid 3D methylcellulose approach to form murine and human ISL1-CPC spheroids that engrafted after myocardial infarction in murine hearts, where they differentiated into cardiomyocytes and endothelial cells, integrating into the myocardium and forming new blood vessels. ISL1-CPC spheroid-treated mice exhibited reduced infarct area and increased blood vessel formation compared with control animals. Moreover, left ventricular (LV) contractile function was significantly better in mice transplanted with ISL1-CPCs 4 weeks after injury than that in control animals. These results provide proof-of-concept of a cardiac repair strategy employing ISL1-CPCs that, based on our previous lineage-tracing studies, are committed to forming heart tissue, in combination with a robust methylcellulose spheroid-based delivery approach.
Figures


Similar articles
-
Transplantation of Isl1+ cardiac progenitor cells in small intestinal submucosa improves infarcted heart function.Stem Cell Res Ther. 2017 Oct 16;8(1):230. doi: 10.1186/s13287-017-0675-2. Stem Cell Res Ther. 2017. PMID: 29037258 Free PMC article.
-
Functional cardiomyocytes derived from Isl1 cardiac progenitors via Bmp4 stimulation.PLoS One. 2014 Dec 18;9(12):e110752. doi: 10.1371/journal.pone.0110752. eCollection 2014. PLoS One. 2014. PMID: 25522363 Free PMC article.
-
Transplantation of multipotent Isl1+ cardiac progenitor cells preserves infarcted heart function in mice.Am J Transl Res. 2017 Mar 15;9(3):1530-1542. eCollection 2017. Am J Transl Res. 2017. PMID: 28386378 Free PMC article.
-
Biology of Isl1+ cardiac progenitor cells in development and disease.Cell Mol Life Sci. 2007 Mar;64(6):674-82. doi: 10.1007/s00018-007-6520-5. Cell Mol Life Sci. 2007. PMID: 17380308 Free PMC article. Review.
-
Endothelial and cardiac progenitor cells for cardiovascular repair: A controversial paradigm in cell therapy.Pharmacol Ther. 2018 Jan;181:156-168. doi: 10.1016/j.pharmthera.2017.08.004. Epub 2017 Aug 19. Pharmacol Ther. 2018. PMID: 28827151 Review.
Cited by
-
The benign nature and rare occurrence of cardiac myxoma as a possible consequence of the limited cardiac proliferative/ regenerative potential: a systematic review.BMC Cancer. 2023 Dec 18;23(1):1245. doi: 10.1186/s12885-023-11723-3. BMC Cancer. 2023. PMID: 38110859 Free PMC article.
-
Cardiac Progenitors Induced from Human Induced Pluripotent Stem Cells with Cardiogenic Small Molecule Effectively Regenerate Infarcted Hearts and Attenuate Fibrosis.Shock. 2018 Dec;50(6):627-639. doi: 10.1097/SHK.0000000000001133. Shock. 2018. PMID: 29485473 Free PMC article.
-
Cardiac stem cells for myocardial regeneration: promising but not ready for prime time.Curr Opin Biotechnol. 2017 Oct;47:30-35. doi: 10.1016/j.copbio.2017.05.009. Epub 2017 Jun 4. Curr Opin Biotechnol. 2017. PMID: 28591641 Free PMC article. Review.
-
microRNAs and cardiac stem cells in heart development and disease.Drug Discov Today. 2019 Jan;24(1):233-240. doi: 10.1016/j.drudis.2018.05.032. Epub 2018 May 28. Drug Discov Today. 2019. PMID: 29852125 Free PMC article. Review.
-
Smyd1 Orchestrates Early Heart Development Through Positive and Negative Gene Regulation.Front Cell Dev Biol. 2021 Apr 1;9:654682. doi: 10.3389/fcell.2021.654682. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33869215 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

