(BB)-Carboryne Complex of Ruthenium: Synthesis by Double B-H Activation at a Single Metal Center
- PMID: 27526855
- PMCID: PMC4999960
- DOI: 10.1021/jacs.6b05172
(BB)-Carboryne Complex of Ruthenium: Synthesis by Double B-H Activation at a Single Metal Center
Abstract
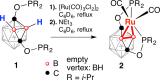
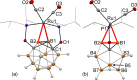
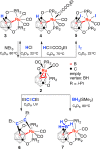
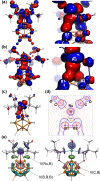
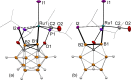
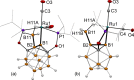
The first example of a transition metal (BB)-carboryne complex containing two boron atoms of the icosahedral cage connected to a single exohedral metal center (POBBOP)Ru(CO)2 (POBBOP = 1,7-OP(i-Pr)2-2,6-dehydro-m-carborane) was synthesized by double B-H activation within the strained m-carboranyl pincer framework. Theoretical calculations revealed that the unique three-membered (BB)>Ru metalacycle is formed by two bent B-Ru σ-bonds with the concomitant increase of the bond order between the two metalated boron atoms. The reactivity of the highly strained electron-rich (BB)-carboryne fragment with small molecules was probed by reactions with electrophiles. The carboryne-carboranyl transformations reported herein represent a new mode of cooperative metal-ligand reactivity of boron-based complexes.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Transition metal-carboryne complexes: synthesis, bonding, and reactivity.Acc Chem Res. 2011 Apr 19;44(4):299-309. doi: 10.1021/ar100156f. Epub 2011 Mar 11. Acc Chem Res. 2011. PMID: 21395260 Review.
-
Expansion of the (BB) 〉metallacycle with coinage metal cations: formation of B-M-Ru-B (M = Cu, Ag, Au) dimetalacyclodiboryls.Chem Sci. 2018 Feb 5;9(9):2601-2608. doi: 10.1039/c8sc00190a. eCollection 2018 Mar 7. Chem Sci. 2018. PMID: 29675252 Free PMC article.
-
Bimetallic Ru-Pd and Trimetallic Ru-Pd-Cu Assemblies on the Carborane Cluster Surface.Inorg Chem. 2021 Nov 15;60(22):16911-16916. doi: 10.1021/acs.inorgchem.1c02799. Epub 2021 Oct 28. Inorg Chem. 2021. PMID: 34710327
-
Rapid reversible borane to boryl hydride exchange by metal shuttling on the carborane cluster surface.Chem Sci. 2017 Aug 1;8(8):5399-5407. doi: 10.1039/c7sc01846k. Epub 2017 May 25. Chem Sci. 2017. PMID: 28970919 Free PMC article.
-
Metal-catalyzed silylation of sp3C-H bonds.Chem Soc Rev. 2021 Apr 26;50(8):5062-5085. doi: 10.1039/d0cs01392g. Chem Soc Rev. 2021. PMID: 33629997 Review.
Cited by
-
Effect of Planarity of Aromatic Rings Appended to o-Carborane on Photophysical Properties: A Series of o-Carboranyl Compounds Based on 2-Phenylpyridine- and 2-(Benzo[b]thiophen-2-yl)pyridine.Molecules. 2019 Jan 7;24(1):201. doi: 10.3390/molecules24010201. Molecules. 2019. PMID: 30621119 Free PMC article.
-
Electrochemical B-H Nitrogenation: Access to Amino Acid and BODIPY-Labeled nido-Carboranes.Angew Chem Int Ed Engl. 2021 Jan 18;60(3):1482-1487. doi: 10.1002/anie.202012105. Epub 2020 Nov 17. Angew Chem Int Ed Engl. 2021. PMID: 32991021 Free PMC article.
-
Electrooxidative o-carborane chalcogenations without directing groups: cage activation by copper catalysis at room temperature.Chem Sci. 2021 Sep 1;12(39):12971-12976. doi: 10.1039/d1sc02905c. eCollection 2021 Oct 13. Chem Sci. 2021. PMID: 34745527 Free PMC article.
-
Iridium Complexes of a Bis(N-pyrrolyl)boryl/Bis(phosphine) PBP Pincer Ligand.Inorg Chem. 2024 Dec 23;63(51):24133-24140. doi: 10.1021/acs.inorgchem.4c03554. Epub 2024 Dec 9. Inorg Chem. 2024. PMID: 39652082 Free PMC article.
-
Rhodium catalyzed cascade cyclization featuring B-H and C-H activation: one-step construction of carborane-fused N-polyheterocycles.Chem Sci. 2018 Jun 29;9(30):6390-6394. doi: 10.1039/c8sc01568f. eCollection 2018 Aug 14. Chem Sci. 2018. PMID: 30310567 Free PMC article.
References
-
- Selander N.; Szabó K. J. Chem. Rev. 2011, 111, 2048.10.1021/cr1002112. - DOI - PubMed
- Choi J.; MacArthur A. H. R.; Brookhart M.; Goldman A. S. Chem. Rev. 2011, 111, 1761.10.1021/cr1003503. - DOI - PubMed
- Gunanathan C.; Milstein D. Chem. Rev. 2014, 114, 12024.10.1021/cr5002782. - DOI - PubMed
- Adhikary A.; Guan H. ACS Catal. 2015, 5, 6858.10.1021/acscatal.5b01688. - DOI
-
- Gusev D. G.; Lough A. J. Organometallics 2002, 21, 2601.10.1021/om020355m. - DOI
- Whited M. T.; Grubbs R. H. Acc. Chem. Res. 2009, 42, 1607.10.1021/ar900103e. - DOI - PubMed
- Doyle L. E.; Piers W. E.; Borau-Garcia J. J. Am. Chem. Soc. 2015, 137, 2187.10.1021/ja512602m. - DOI - PubMed
- Comanescu C. C.; Iluc V. M. Organometallics 2014, 33, 6059.10.1021/om500682s. - DOI
- Liang L. C. Coord. Chem. Rev. 2006, 250, 1152.10.1016/j.ccr.2006.01.001. - DOI
- Morgan E.; MacLean D. F.; McDonald R.; Turculet L. J. Am. Chem. Soc. 2009, 131, 14234.10.1021/ja906646v. - DOI - PubMed
- Caulton K. G. Eur. J. Inorg. Chem. 2012, 2012, 435.10.1002/ejic.201100623. - DOI
-
- Dang L.; Lin Z.; Marder T. B. Chem. Commun. 2009, 27, 3987.10.1039/b903098k. - DOI - PubMed
- Segawa Y.; Yamashita M.; Nozaki K. J. Am. Chem. Soc. 2009, 131, 9201.10.1021/ja9037092. - DOI - PubMed
- Chaplin A. B.; Weller A. S. Angew. Chem., Int. Ed. 2010, 49, 581.10.1002/anie.200905185. - DOI - PubMed
- Harman W. H.; Peters J. C. J. Am. Chem. Soc. 2012, 134, 5080.10.1021/ja211419t. - DOI - PubMed
- Lin T.-P.; Peters J. C. J. Am. Chem. Soc. 2013, 135, 15310.10.1021/ja408397v. - DOI - PubMed
- Bontemps S.; Gornitzka H.; Bouhadir G.; Miqueu K.; Bourissou D. Angew. Chem., Int. Ed. 2006, 45, 1611.10.1002/anie.200503649. - DOI - PubMed
- Bontemps S.; Bouhadir G.; Gu W.; Mercy M.; Chen C.-H.; Foxman B. M.; Maron L.; Ozerov O. V.; Bourissou D. Angew. Chem., Int. Ed. 2008, 47, 1481.10.1002/anie.200705024. - DOI - PubMed
- Braunschweig H.; Dewhurst R. D. Dalton Trans. 2010, 40, 549.10.1039/C0DT01181A. - DOI - PubMed
- Braunschweig H.; Dewhurst R. D.; Schneider A. Chem. Rev. 2010, 110, 3924.10.1021/cr900333n. - DOI - PubMed
- Shih W.-C.; Gu W.; MacInnis M. C.; Timpa S. D.; Bhuvanesh N.; Zhou J.; Ozerov O. V. J. Am. Chem. Soc. 2016, 138, 2086.10.1021/jacs.5b11706. - DOI - PubMed
- Hill A. F.; Lee S. B.; Park J.; Shang R.; Willis A. C. Organometallics 2010, 29, 5661.10.1021/om100557q. - DOI
- Hill A. F.; McQueen C. M. A. Organometallics 2014, 33, 1977.10.1021/om5001106. - DOI
- Owen G. R. Chem. Soc. Rev. 2012, 41, 3535.10.1039/c2cs15346g. - DOI - PubMed
- Owen G. R. Chem. Commun. 2016, 10.1039/C6CC03817D. - DOI - PubMed
-
- Grimes R. N. Dalton Trans. 2015, 44, 5939.10.1039/C5DT00231A. - DOI - PubMed
- Popescu A. R.; Teixidor F.; Viñas C. Coord. Chem. Rev. 2014, 269, 54.10.1016/j.ccr.2014.02.016. - DOI
- Yinghuai Z.; Hosmane N. S. J. Organomet. Chem. 2013, 747, 25.10.1016/j.jorganchem.2013.03.042. - DOI
- Yao Z.-J.; Jin G.-X. Coord. Chem. Rev. 2013, 257, 2522.10.1016/j.ccr.2013.02.004. - DOI
- Douvris C.; Michl J. Chem. Rev. 2013, 113, PR179.10.1021/cr400059k. - DOI - PubMed
- Yamashita M. Bull. Chem. Soc. Jpn. 2015, 89, 269.10.1246/bcsj.20150355. - DOI
- Gleeson B.; Carroll P. J.; Sneddon L. G. J. Am. Chem. Soc. 2013, 135, 12407.10.1021/ja405977q. - DOI - PubMed
- Teixidor F.; Flores M. A.; Viñas C.; Sillanpää R.; Kivekäs R. J. Am. Chem. Soc. 2000, 122, 1963.10.1021/ja992970w. - DOI
- El-Hellani A.; Kefalidis C. E.; Tham F. S.; Maron L.; Lavallo V. Organometallics 2013, 32, 6887.10.1021/om401001p. - DOI
- Spokoyny A. M.; Reuter M. G.; Stern C. L.; Ratner M. A.; Seideman T.; Mirkin C. A. J. Am. Chem. Soc. 2009, 131, 9482.10.1021/ja902526k. - DOI - PMC - PubMed
- Kirchmann M.; Wesemann L. Dalton Trans. 2008, 4, 444.10.1039/B715305H. - DOI - PubMed
- Himmelspach A.; Finze M.; Raub S. Angew. Chem., Int. Ed. 2011, 50, 2628.10.1002/anie.201007239. - DOI - PubMed
- El-Zaria M. E.; Arii H.; Nakamura H. Inorg. Chem. 2011, 50, 4149.10.1021/ic2002095. - DOI - PubMed
- Lavallo V.; Wright J. H.; Tham F. S.; Quinlivan S. Angew. Chem., Int. Ed. 2013, 52, 3172.10.1002/anie.201209107. - DOI - PubMed
- Tsang M. Y.; Viñas C.; Teixidor F.; Planas J. G.; Conde N.; SanMartin R.; Herrero M. T.; Domínguez E.; Lledós A.; Vidossich P.; Choquesillo-Lazarte D. Inorg. Chem. 2014, 53, 9284.10.1021/ic5013999. - DOI - PubMed
- Eleazer B. J.; Smith M. D.; Peryshkov D. V. Organometallics 2016, 35, 106.10.1021/acs.organomet.5b00807. - DOI
- Spokoyny A. M. Pure Appl. Chem. 2013, 85, 903.10.1351/PAC-CON-13-01-13. - DOI - PMC - PubMed
-
-
For selected recent examples and reviews, see:
- Núñez R.; Romero I.; Teixidor F.; Viñas C. Chem. Soc. Rev. 2016, 10.1039/C6CS00159A. - DOI - PubMed
- Wee K.-R.; Cho Y.-J.; Jeong S.; Kwon S.; Lee J.-D.; Suh I.-H.; Kang S. O. J. Am. Chem. Soc. 2012, 134, 17982.10.1021/ja3066623. - DOI - PubMed
- Koshino M.; Tanaka T.; Solin N.; Suenaga K.; Isobe H.; Nakamura E. Science 2007, 316, 853.10.1126/science.1138690. - DOI - PubMed
- Schwartz J. J.; Mendoza M. A.; Wattanatorn N.; Zhao Y.; Nguyen V. T.; Spokoyny A. M.; Mirkin C. A.; Baše T.; Weiss P. S. J. Am. Chem. Soc. 2016, 138, 5957.10.1021/jacs.6b02026. - DOI - PubMed
- Kirlikovali K. O.; Axtell J. A.; Gonzalez A.; Phung A. C.; Khan S. I.; Spokoyny A. M. Chem. Sci. 2016, 7, 5132.10.1039/C6SC01146B. - DOI - PMC - PubMed
- Tutusaus O.; Mohtadi R.; Arthur T. S.; Mizuno F.; Nelson E. G.; Sevryugina Y. V. Angew. Chem., Int. Ed. 2015, 54, 7900.10.1002/anie.201412202. - DOI - PubMed
- Scholz M.; Hey-Hawkins E. Chem. Rev. 2011, 111, 7035.10.1021/cr200038x. - DOI - PubMed
- McArthur S. G.; Geng L.; Guo J.; Lavallo V. Inorg. Chem. Front. 2015, 2, 1101.10.1039/C5QI00171D. - DOI
- Kracke G. R.; VanGordon M. R.; Sevryugina Y. V.; Kueffer P. J.; Kabytaev K.; Jalisatgi S. S.; Hawthorne M. F. ChemMedChem 2015, 10, 62.10.1002/cmdc.201402369. - DOI - PubMed
- Kaszynski P. Collect. Czech. Chem. Commun. 1999, 64, 895.10.1135/cccc19990895. - DOI
- Dash B. P.; Satapathy R.; Maguire J. A.; Hosmane N. S. New J. Chem. 2011, 35, 1955.10.1039/c1nj20228f. - DOI
-
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

