Lipid somersaults: Uncovering the mechanisms of protein-mediated lipid flipping
- PMID: 27528189
- PMCID: PMC5127727
- DOI: 10.1016/j.plipres.2016.08.003
Lipid somersaults: Uncovering the mechanisms of protein-mediated lipid flipping
Abstract
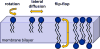
Membrane lipids diffuse rapidly in the plane of the membrane but their ability to flip spontaneously across a membrane bilayer is hampered by a significant energy barrier. Thus spontaneous flip-flop of polar lipids across membranes is very slow, even though it must occur rapidly to support diverse aspects of cellular life. Here we discuss the mechanisms by which rapid flip-flop occurs, and what role lipid flipping plays in membrane homeostasis and cell growth. We focus on conceptual aspects, highlighting mechanistic insights from biochemical and in silico experiments, and the recent, ground-breaking identification of a number of lipid scramblases.
Keywords: Flippase; Floppase; Membrane asymmetry; Phosphatidylserine; Photoreceptor; Scramblase.
Copyright © 2016 Elsevier B.V. All rights reserved.
Figures




Similar articles
-
Opsin is a phospholipid flippase.Curr Biol. 2011 Jan 25;21(2):149-53. doi: 10.1016/j.cub.2010.12.031. Epub 2011 Jan 13. Curr Biol. 2011. PMID: 21236677 Free PMC article.
-
The role of lipid scramblases in regulating lipid distributions at cellular membranes.Biochem Soc Trans. 2023 Oct 31;51(5):1857-1869. doi: 10.1042/BST20221455. Biochem Soc Trans. 2023. PMID: 37767549 Review.
-
Flipping and flopping--lipids on the move.IUBMB Life. 2011 Sep;63(9):736-46. doi: 10.1002/iub.515. Epub 2011 Jul 26. IUBMB Life. 2011. PMID: 21793163 Review.
-
The Ins and Outs of Lipid Flip-Flop.Acc Chem Res. 2017 Jan 17;50(1):58-65. doi: 10.1021/acs.accounts.6b00435. Epub 2016 Dec 13. Acc Chem Res. 2017. PMID: 27959517
-
Defining key roles for auxiliary proteins in an ABC transporter that maintains bacterial outer membrane lipid asymmetry.Elife. 2016 Aug 16;5:e19042. doi: 10.7554/eLife.19042. Elife. 2016. PMID: 27529189 Free PMC article.
Cited by
-
Development of membrane-insertable lipid scrambling peptides: A time-resolved small-angle neutron scattering study.Struct Dyn. 2021 Mar 19;8(2):024301. doi: 10.1063/4.0000045. eCollection 2021 Mar. Struct Dyn. 2021. PMID: 33758768 Free PMC article.
-
FTY720 induces non-canonical phosphatidylserine externalization and cell death in acute myeloid leukemia.Cell Death Dis. 2019 Nov 7;10(11):847. doi: 10.1038/s41419-019-2080-5. Cell Death Dis. 2019. PMID: 31699964 Free PMC article.
-
Gain-of-Function Mutations in the Phospholipid Flippase MprF Confer Specific Daptomycin Resistance.mBio. 2018 Dec 18;9(6):e01659-18. doi: 10.1128/mBio.01659-18. mBio. 2018. PMID: 30563904 Free PMC article.
-
C. elegans TAT-6, a putative aminophospholipid translocase, is expressed in sujc cells in the hermaphrodite gonad.MicroPubl Biol. 2021 Nov 4;2021:10.17912/micropub.biology.000495. doi: 10.17912/micropub.biology.000495. eCollection 2021. MicroPubl Biol. 2021. PMID: 34746684 Free PMC article.
-
Phospholipid subcellular localization and dynamics.J Biol Chem. 2018 Apr 27;293(17):6230-6240. doi: 10.1074/jbc.R117.000582. Epub 2018 Mar 27. J Biol Chem. 2018. PMID: 29588369 Free PMC article. Review.
References
-
- Zwaal RFA, Demel RA, Roelofsen B, van Deenen LLM. The lipid bilayer concept of cell membranes. Trends in Biochemical Sciences. 1976;1:112–4.
-
- Tanford C. The hydrophobic effect: formation of micelles and biological membranes. New York: John Wiley & Sons; 1973.
-
- Mouritsen OG. Life - As a Matter of Fat. Berlin Heidelberg: GmbH & Co K.: Springer-Verlag; 2005.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

