Defining key roles for auxiliary proteins in an ABC transporter that maintains bacterial outer membrane lipid asymmetry
- PMID: 27529189
- PMCID: PMC5016091
- DOI: 10.7554/eLife.19042
Defining key roles for auxiliary proteins in an ABC transporter that maintains bacterial outer membrane lipid asymmetry
Abstract
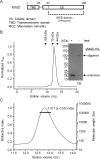
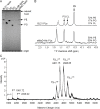
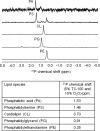
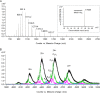
In Gram-negative bacteria, lipid asymmetry is critical for the function of the outer membrane (OM) as a selective permeability barrier, but how it is established and maintained is poorly understood. Here, we characterize a non-canonical ATP-binding cassette (ABC) transporter in Escherichia coli that provides energy for maintaining OM lipid asymmetry via the transport of aberrantly localized phospholipids (PLs) from the OM to the inner membrane (IM). We establish that the transporter comprises canonical components, MlaF and MlaE, and auxiliary proteins, MlaD and MlaB, of previously unknown functions. We further demonstrate that MlaD forms extremely stable hexamers within the complex, functions in substrate binding with strong affinity for PLs, and modulates ATP hydrolytic activity. In addition, MlaB plays critical roles in both the assembly and activity of the transporter. Our work provides mechanistic insights into how the MlaFEDB complex participates in ensuring active retrograde PL transport to maintain OM lipid asymmetry.
Keywords: E. coli; biochemistry; infectious disease; intermembrane lipid transport; mammalian cell entry (MCE) domain; microbiology; phospholipid binding; protein complex assembly; sulfate transport and anti-sigma factor antagonist (STAS) domain.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures






Similar articles
-
ATP disrupts lipid-binding equilibrium to drive retrograde transport critical for bacterial outer membrane asymmetry.Proc Natl Acad Sci U S A. 2021 Dec 14;118(50):e2110055118. doi: 10.1073/pnas.2110055118. Proc Natl Acad Sci U S A. 2021. PMID: 34873038 Free PMC article.
-
MCE domain proteins: conserved inner membrane lipid-binding proteins required for outer membrane homeostasis.Sci Rep. 2017 Aug 17;7(1):8608. doi: 10.1038/s41598-017-09111-6. Sci Rep. 2017. PMID: 28819315 Free PMC article.
-
Outer membrane lipid homeostasis via retrograde phospholipid transport in Escherichia coli.Mol Microbiol. 2017 Nov;106(3):395-408. doi: 10.1111/mmi.13772. Epub 2017 Sep 6. Mol Microbiol. 2017. PMID: 28815827
-
Current mechanistic understanding of intermembrane lipid trafficking important for maintenance of bacterial outer membrane lipid asymmetry.Curr Opin Chem Biol. 2021 Dec;65:163-171. doi: 10.1016/j.cbpa.2021.09.004. Epub 2021 Nov 6. Curr Opin Chem Biol. 2021. PMID: 34753108 Review.
-
Structure and mechanism of the bacterial lipid ABC transporter, MlaFEDB.Curr Opin Struct Biol. 2022 Oct;76:102429. doi: 10.1016/j.sbi.2022.102429. Epub 2022 Aug 15. Curr Opin Struct Biol. 2022. PMID: 35981415 Free PMC article. Review.
Cited by
-
Assembly and Maintenance of Lipids at the Bacterial Outer Membrane.Chem Rev. 2021 May 12;121(9):5098-5123. doi: 10.1021/acs.chemrev.0c00587. Epub 2020 Sep 21. Chem Rev. 2021. PMID: 32955879 Free PMC article. Review.
-
Salmonella enterica Serovar Typhimurium Uses PbgA/YejM To Regulate Lipopolysaccharide Assembly during Bacteremia.Infect Immun. 2019 Dec 17;88(1):e00758-19. doi: 10.1128/IAI.00758-19. Print 2019 Dec 17. Infect Immun. 2019. PMID: 31611279 Free PMC article.
-
Salmonella Tol-Pal Reduces Outer Membrane Glycerophospholipid Levels for Envelope Homeostasis and Survival during Bacteremia.Infect Immun. 2018 Jun 21;86(7):e00173-18. doi: 10.1128/IAI.00173-18. Print 2018 Jul. Infect Immun. 2018. PMID: 29735519 Free PMC article.
-
The Escherichia coli Phospholipase PldA Regulates Outer Membrane Homeostasis via Lipid Signaling.mBio. 2018 Mar 20;9(2):e00379-18. doi: 10.1128/mBio.00379-18. mBio. 2018. PMID: 29559571 Free PMC article.
-
Direct visualization of a molecular handshake that governs kin recognition and tissue formation in myxobacteria.Nat Commun. 2019 Jul 12;10(1):3073. doi: 10.1038/s41467-019-11108-w. Nat Commun. 2019. PMID: 31300643 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

