Dual Anti-Inflammatory and Anti-Angiogenic Action of miR-15a in Diabetic Retinopathy
- PMID: 27531575
- PMCID: PMC5049929
- DOI: 10.1016/j.ebiom.2016.08.013
Dual Anti-Inflammatory and Anti-Angiogenic Action of miR-15a in Diabetic Retinopathy
Abstract
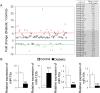
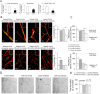
Activation of pro-inflammatory and pro-angiogenic pathways in the retina and the bone marrow contributes to pathogenesis of diabetic retinopathy. We identified miR-15a as key regulator of both pro-inflammatory and pro-angiogenic pathways through direct binding and inhibition of the central enzyme in the sphingolipid metabolism, ASM, and the pro-angiogenic growth factor, VEGF-A. miR-15a was downregulated in diabetic retina and bone marrow cells. Over-expression of miR-15a downregulated, and inhibition of miR-15a upregulated ASM and VEGF-A expression in retinal cells. In addition to retinal effects, migration and retinal vascular repair function was impaired in miR-15a inhibitor-treated circulating angiogenic cells (CAC). Diabetic mice overexpressing miR-15a under Tie-2 promoter had normalized retinal permeability compared to wild type littermates. Importantly, miR-15a overexpression led to modulation toward nondiabetic levels, rather than complete inhibition of ASM and VEGF-A providing therapeutic effect without detrimental consequences of ASM and VEGF-A deficiencies.
Keywords: Diabetic retinopathy; Dyslipidemias; Sphingolipids; Vascular system injuries; microRNA.
Copyright © 2016 Forschungsgesellschaft für Arbeitsphysiologie und Arbeitschutz e.V. Published by Elsevier B.V. All rights reserved.
Figures




Similar articles
-
Role of Acid Sphingomyelinase in Shifting the Balance Between Proinflammatory and Reparative Bone Marrow Cells in Diabetic Retinopathy.Stem Cells. 2016 Apr;34(4):972-83. doi: 10.1002/stem.2259. Epub 2016 Jan 4. Stem Cells. 2016. PMID: 26676316 Free PMC article.
-
Upregulated VEGF and Robo4 correlate with the reduction of miR-15a in the development of diabetic retinopathy.Endocrine. 2019 Jul;65(1):35-45. doi: 10.1007/s12020-019-01921-0. Epub 2019 Apr 12. Endocrine. 2019. PMID: 30980286 Free PMC article.
-
miR-15a and miR-16 affect the angiogenesis of multiple myeloma by targeting VEGF.Carcinogenesis. 2013 Feb;34(2):426-35. doi: 10.1093/carcin/bgs333. Epub 2012 Oct 26. Carcinogenesis. 2013. PMID: 23104180
-
Neurovascular regulation in diabetic retinopathy and emerging therapies.Cell Mol Life Sci. 2021 Aug;78(16):5977-5985. doi: 10.1007/s00018-021-03893-9. Epub 2021 Jul 7. Cell Mol Life Sci. 2021. PMID: 34230991 Free PMC article. Review.
-
The effects of microRNAs in activating neovascularization pathways in diabetic retinopathy.J Cell Biochem. 2019 Jun;120(6):9514-9521. doi: 10.1002/jcb.28227. Epub 2018 Dec 16. J Cell Biochem. 2019. PMID: 30556195 Review.
Cited by
-
Potential Role of miRNAs as Theranostic Biomarkers of Epilepsy.Mol Ther Nucleic Acids. 2018 Dec 7;13:275-290. doi: 10.1016/j.omtn.2018.09.008. Epub 2018 Sep 13. Mol Ther Nucleic Acids. 2018. PMID: 30321815 Free PMC article. Review.
-
miR-15a/16 reduces retinal leukostasis through decreased pro-inflammatory signaling.J Neuroinflammation. 2016 Dec 8;13(1):305. doi: 10.1186/s12974-016-0771-8. J Neuroinflammation. 2016. PMID: 27931222 Free PMC article.
-
Hematopoietic stem/progenitor involvement in retinal microvascular repair during diabetes: Implications for bone marrow rejuvenation.Vision Res. 2017 Oct;139:211-220. doi: 10.1016/j.visres.2017.06.016. Epub 2017 Oct 31. Vision Res. 2017. PMID: 29042190 Free PMC article. Review.
-
The Mechanism of Diabetic Retinopathy Pathogenesis Unifying Key Lipid Regulators, Sirtuin 1 and Liver X Receptor.EBioMedicine. 2017 Aug;22:181-190. doi: 10.1016/j.ebiom.2017.07.008. Epub 2017 Jul 11. EBioMedicine. 2017. PMID: 28774737 Free PMC article.
-
miR-15a/16 inhibits TGF-beta3/VEGF signaling and increases retinal endothelial cell barrier proteins.Vision Res. 2017 Oct;139:23-29. doi: 10.1016/j.visres.2017.07.007. Epub 2017 Aug 7. Vision Res. 2017. PMID: 28774775 Free PMC article.
References
-
- Abu El-Asrar A.M., Struyf S., Verbeke H., Van Damme J., Geboes K. Circulating bone-marrow-derived endothelial precursor cells contribute to neovascularization in diabetic epiretinal membranes. Acta Ophthalmol. 2011;89:222–228. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

