Absence of CCR2 results in an inflammaging environment in young mice with age-independent impairments in muscle regeneration
- PMID: 27531927
- PMCID: PMC5069084
- DOI: 10.1189/jlb.3MA0316-104R
Absence of CCR2 results in an inflammaging environment in young mice with age-independent impairments in muscle regeneration
Abstract
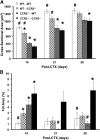
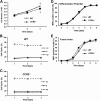
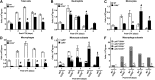
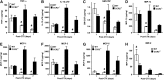
Skeletal muscle regeneration requires coordination between dynamic cellular populations and tissue microenvironments. Macrophages, recruited via CCR2, are essential for regeneration; however, the contribution of macrophages and the role of CCR2 on nonhematopoietic cells has not been defined. In addition, aging and sex interactions in regeneration and sarcopenia are unclear. Muscle regeneration was measured in young (3-6 mo), middle (11-15 mo), old (24-32 mo) male and female CCR2-/- mice. Whereas age-related muscle atrophy/sarcopenia was present, regenerated myofiber cross-sectional area (CSA) in CCR2-/- mice was comparably impaired across all ages and sexes, with increased adipocyte area compared with wild-type (WT) mice. CCR2-/- mice myofibers achieved approximately one third of baseline CSA even 84 d after injury. Regenerated CSA and clearance of necrotic tissue were dependent on bone marrow-derived cellular expression of CCR2. Myogenic progenitor cells isolated from WT and CCR2-/- mice exhibited comparable proliferation and differentiation capacity. The most striking cellular anomaly in injured muscle of CCR2-/- mice was markedly decreased macrophages, with a predominance of Ly6C- anti-inflammatory monocytes/macrophages. Ablation of proinflammatory TLR signaling did not affect muscle regeneration or resolution of necrosis. Of interest, many proinflammatory, proangiogenic, and chemotactic cytokines were markedly elevated in injured muscle of CCR2-/- relative to WT mice despite impairments in macrophage recruitment. Collectively, these results suggest that CCR2 on bone marrow-derived cells, likely macrophages, were essential to muscle regeneration independent of TLR signaling, aging, and sex. Decreased proinflammatory monocytes/macrophages actually promoted a proinflammatory microenvironment, which suggests that inflammaging was present in young CCR2-/- mice.
Keywords: TLRs; monocytes/macrophages; myogenic progenitor cells; sarcopenia.
© Society for Leukocyte Biology.
Figures


References
-
- Martinez C. O., McHale M. J., Wells J. T., Ochoa O., Michalek J. E., McManus L. M., Shireman P. K. (2010) Regulation of skeletal muscle regeneration by CCR2-activating chemokines is directly related to macrophage recruitment. Am. J. Physiol. Regul. Integr. Comp. Physiol. 299, R832–R842. - PMC - PubMed
-
- Ochoa O., Sun D., Reyes-Reyna S. M., Waite L. L., Michalek J. E., McManus L. M., Shireman P. K. (2007) Delayed angiogenesis and VEGF production in CCR2-/- mice during impaired skeletal muscle regeneration. Am. J. Physiol. Regul. Integr. Comp. Physiol. 293, R651–R661. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

