Specific polyunsaturated fatty acids modulate lipid delivery and oocyte development in C. elegans revealed by molecular-selective label-free imaging
- PMID: 27535493
- PMCID: PMC4989181
- DOI: 10.1038/srep32021
Specific polyunsaturated fatty acids modulate lipid delivery and oocyte development in C. elegans revealed by molecular-selective label-free imaging
Abstract
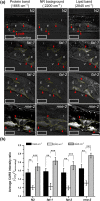
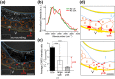
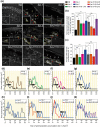
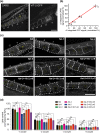
Polyunsaturated fatty acids (PUFAs) exhibit critical functions in biological systems and their importance during animal oocyte maturation has been increasingly recognized. However, the detailed mechanism of lipid transportation for oocyte development remains largely unknown. In this study, the transportation of yolk lipoprotein (lipid carrier) and the rate of lipid delivery into oocytes in live C. elegans were examined for the first time by using coherent anti-Stokes Raman scattering (CARS) microscopy. The accumulation of secreted yolk lipoprotein in the pseudocoelom of live C. elegans can be detected by CARS microscopy at both protein (~1665 cm(-1)) and lipid (~2845 cm(-1)) Raman bands. In addition, an image analysis protocol was established to quantitatively measure the levels of secreted yolk lipoprotein aberrantly accumulated in PUFA-deficient fat mutants (fat-1, fat-2, fat-3, fat-4) and PUFA-supplemented fat-2 worms (the PUFA add-back experiments). Our results revealed that the omega-6 PUFAs, not omega-3 PUFAs, play a critical role in modulating lipid/yolk level in the oocytes and regulating reproductive efficiency of C. elegans. This work demonstrates the value of using CARS microscopy as a molecular-selective label-free imaging technique for the study of PUFA regulation and oocyte development in C. elegans.
Figures

Similar articles
-
Lipid droplet pattern and nondroplet-like structure in two fat mutants of Caenorhabditis elegans revealed by coherent anti-Stokes Raman scattering microscopy.J Biomed Opt. 2014 Jan;19(1):011011. doi: 10.1117/1.JBO.19.1.011011. J Biomed Opt. 2014. PMID: 23979461
-
Polyunsaturated fatty acids are involved in regulatory mechanism of fatty acid homeostasis via daf-2/insulin signaling in Caenorhabditis elegans.Mol Cell Endocrinol. 2010 Jul 29;323(2):183-92. doi: 10.1016/j.mce.2010.03.004. Epub 2010 Mar 11. Mol Cell Endocrinol. 2010. PMID: 20226839
-
The cytochrome b5 reductase HPO-19 is required for biosynthesis of polyunsaturated fatty acids in Caenorhabditis elegans.Biochim Biophys Acta. 2016 Apr;1861(4):310-9. doi: 10.1016/j.bbalip.2016.01.009. Epub 2016 Jan 20. Biochim Biophys Acta. 2016. PMID: 26806391
-
Fatty Acid Desaturases, Polyunsaturated Fatty Acid Regulation, and Biotechnological Advances.Nutrients. 2016 Jan 4;8(1):23. doi: 10.3390/nu8010023. Nutrients. 2016. PMID: 26742061 Free PMC article. Review.
-
Synthesis and function of fatty acids and oxylipins, with a focus on Caenorhabditis elegans.Prostaglandins Other Lipid Mediat. 2020 Jun;148:106426. doi: 10.1016/j.prostaglandins.2020.106426. Epub 2020 Feb 4. Prostaglandins Other Lipid Mediat. 2020. PMID: 32032704 Review.
Cited by
-
Reproductive Suicide: Similar Mechanisms of Aging in C. elegans and Pacific Salmon.Front Cell Dev Biol. 2021 Aug 27;9:688788. doi: 10.3389/fcell.2021.688788. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34513830 Free PMC article.
-
Perfluorooctanesulfonic acid (PFOS) and perfluorobutanesulfonic acid (PFBS) impaired reproduction and altered offspring physiological functions in Caenorhabditis elegans.Food Chem Toxicol. 2020 Nov;145:111695. doi: 10.1016/j.fct.2020.111695. Epub 2020 Aug 22. Food Chem Toxicol. 2020. PMID: 32835727 Free PMC article.
-
FORWARD GENETICS IN C. ELEGANS REVEALS GENETIC ADAPTATIONS TO POLYUNSATURATED FATTY ACID DEFICIENCY.bioRxiv [Preprint]. 2025 Jun 5:2024.11.08.622646. doi: 10.1101/2024.11.08.622646. bioRxiv. 2025. Update in: Elife. 2025 Jul 08;13:RP104181. doi: 10.7554/eLife.104181. PMID: 40501978 Free PMC article. Updated. Preprint.
-
A genetic titration of membrane composition in Caenorhabditis elegans reveals its importance for multiple cellular and physiological traits.Genetics. 2021 Aug 26;219(1):iyab093. doi: 10.1093/genetics/iyab093. Genetics. 2021. PMID: 34125894 Free PMC article.
-
Identifying lipid particle sub-types in live Caenorhabditis elegans with two-photon fluorescence lifetime imaging.Front Chem. 2023 Apr 13;11:1161775. doi: 10.3389/fchem.2023.1161775. eCollection 2023. Front Chem. 2023. PMID: 37123874 Free PMC article.
References
-
- Dunning K. R., Russell D. L. & Robker R. L. Lipids and oocyte developmental competence: the role of fatty acids and beta-oxidation. Reproduction 148, R15–R27 (2014). - PubMed
-
- Aguilar P. S. & de Mendoza D. Control of fatty acid desaturation: a mechanism conserved from bacteria to humans. Mol Microbiol. 62, 1507–1514 (2006). - PubMed
-
- Kubagawa H. M. et al.. Oocyte signals derived from polyunsaturated fatty acids control sperm recruitment in vivo. Nat. Cell. Biol. 8, 1143–1183 (2006). - PubMed
-
- Branicky R., Desjardins D., Liu J. L. & Hekimi S. Lipid Transport and Signaling in Caenorhabditis elegans. Dev. Dynam. 239, 1365–1377 (2010). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

