Fibroblast Growth Factor 23 Regulation by Systemic and Local Osteoblast-Synthesized 1,25-Dihydroxyvitamin D
- PMID: 27535551
- PMCID: PMC5280015
- DOI: 10.1681/ASN.2016010066
Fibroblast Growth Factor 23 Regulation by Systemic and Local Osteoblast-Synthesized 1,25-Dihydroxyvitamin D
Abstract
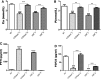
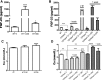
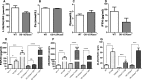
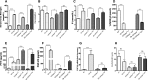
Circulating levels of fibroblast growth factor 23 (FGF23) increase during the early stages of kidney disease, but the underlying mechanism remains incompletely characterized. We investigated the role of vitamin D metabolites in regulating intact FGF23 production in genetically modified mice without and with adenine-induced uremia. Exogenous calcitriol (1,25-dihydroxyvitamin D) and high circulating levels of calcidiol (25-hydroxyvitamin D) each increased serum FGF23 levels in wild-type mice and in mice with global deficiency of the Cyp27b1 gene encoding 25-hydroxyvitamin D 1-α-hydroxylase, which produces 1,25-hydroxyvitamin D. Compared with wild-type mice, normal, or uremic mice lacking Cyp27b1 had lower levels of serum FGF23, despite having high concentrations of parathyroid hormone, but administration of exogenous 1,25-dihydroxyvitamin D increased FGF23 levels. Furthermore, raising serum calcium levels in Cyp27b1-depleted mice directly increased FGF23 levels and indirectly enhanced the action of ambient vitamin D metabolites via the vitamin D receptor. In chromatin immunoprecipitation assays, 25-hydroxyvitamin D promoted binding of the vitamin D receptor and retinoid X receptor to the promoters of osteoblastic target genes. Conditional osteoblastic deletion of Cyp27b1 caused lower serum FGF23 levels, despite normal circulating levels of vitamin D metabolites. In adenine-induced uremia, only a modest increase in serum FGF23 levels occurred in mice with osteoblastic deletion of Cyp27b1 (12-fold) compared with a large increase (58-fold) in wild-type mice. Therefore, in addition to the direct effect of high circulating concentrations of 25-hydroxyvitamin D, local osteoblastic conversion of 25-hydroxyvitamin D to 1,25-dihydroxyvitamin D appears to be an important positive regulator of FGF23 production, particularly in uremia.
Keywords: Activated Vitamin D; FGF23; Vitamin D; calcium; parathyroid hormone.
Copyright © 2017 by the American Society of Nephrology.
Figures


Comment in
-
The Pas de Trois of Vitamin D, FGF23, and PTH.J Am Soc Nephrol. 2017 Feb;28(2):393-395. doi: 10.1681/ASN.2016090944. Epub 2016 Nov 2. J Am Soc Nephrol. 2017. PMID: 27807212 Free PMC article. No abstract available.
Similar articles
-
Fibroblast growth factor 23 inhibits extrarenal synthesis of 1,25-dihydroxyvitamin D in human monocytes.J Bone Miner Res. 2013 Jan;28(1):46-55. doi: 10.1002/jbmr.1740. J Bone Miner Res. 2013. PMID: 22886720 Free PMC article.
-
Bone formation regulates circulating concentrations of fibroblast growth factor 23.Endocrinology. 2009 Nov;150(11):4835-45. doi: 10.1210/en.2009-0472. Epub 2009 Oct 9. Endocrinology. 2009. PMID: 19819968
-
Targeted genomic deletions identify diverse enhancer functions and generate a kidney-specific, endocrine-deficient Cyp27b1 pseudo-null mouse.J Biol Chem. 2019 Jun 14;294(24):9518-9535. doi: 10.1074/jbc.RA119.008760. Epub 2019 May 3. J Biol Chem. 2019. PMID: 31053643 Free PMC article.
-
Mechanistic homeostasis of vitamin D metabolism in the kidney through reciprocal modulation of Cyp27b1 and Cyp24a1 expression.J Steroid Biochem Mol Biol. 2020 Feb;196:105500. doi: 10.1016/j.jsbmb.2019.105500. Epub 2019 Oct 16. J Steroid Biochem Mol Biol. 2020. PMID: 31629064 Free PMC article. Review.
-
Reciprocal control of 1,25-dihydroxyvitamin D and FGF23 formation involving the FGF23/Klotho system.Clin J Am Soc Nephrol. 2010 Sep;5(9):1717-22. doi: 10.2215/CJN.02680310. Epub 2010 Aug 26. Clin J Am Soc Nephrol. 2010. PMID: 20798257 Review.
Cited by
-
High-phosphorus diets reduce aortic lesions and cardiomyocyte size and modify lipid metabolism in Ldl receptor knockout mice.Sci Rep. 2020 Nov 27;10(1):20748. doi: 10.1038/s41598-020-77509-w. Sci Rep. 2020. PMID: 33247205 Free PMC article.
-
Vitamin D Effects on Bone Homeostasis and Cardiovascular System in Patients with Chronic Kidney Disease and Renal Transplant Recipients.Nutrients. 2021 Apr 25;13(5):1453. doi: 10.3390/nu13051453. Nutrients. 2021. PMID: 33922902 Free PMC article. Review.
-
Dysequilibrium of the PTH-FGF23-vitamin D axis in relapsing remitting multiple sclerosis; a longitudinal study.Mol Med. 2018 May 30;24(1):27. doi: 10.1186/s10020-018-0028-3. Mol Med. 2018. PMID: 30134801 Free PMC article.
-
Protective Role of Vitamin D in Renal Tubulopathies.Metabolites. 2020 Mar 19;10(3):115. doi: 10.3390/metabo10030115. Metabolites. 2020. PMID: 32204545 Free PMC article. Review.
-
A kidney-specific genetic control module in mice governs endocrine regulation of the cytochrome P450 gene Cyp27b1 essential for vitamin D3 activation.J Biol Chem. 2017 Oct 20;292(42):17541-17558. doi: 10.1074/jbc.M117.806901. Epub 2017 Aug 14. J Biol Chem. 2017. PMID: 28808057 Free PMC article.
References
-
- ADHR Consortium : Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat Genet 26: 345–348, 2000 - PubMed
-
- Bai XY, Miao D, Goltzman D, Karaplis AC: The autosomal dominant hypophosphatemic rickets R176Q mutation in fibroblast growth factor 23 resists proteolytic cleavage and enhances in vivo biological potency. J Biol Chem 278: 9843–9849, 2003 - PubMed
-
- Larsson T, Marsell R, Schipani E, Ohlsson C, Ljunggren O, Tenenhouse HS, Jüppner H, Jonsson KB: Transgenic mice expressing fibroblast growth factor 23 under the control of the α1(I) collagen promoter exhibit growth retardation, osteomalacia, and disturbed phosphate homeostasis. Endocrinology 145: 3087–3094, 2004 - PubMed
-
- Shimada T, Urakawa I, Yamazaki Y, Hasegawa H, Hino R, Yoneya T, Takeuchi Y, Fujita T, Fukumoto S, Yamashita T: FGF-23 transgenic mice demonstrate hypophosphatemic rickets with reduced expression of sodium phosphate cotransporter type IIa. Biochem Biophys Res Commun 314: 409–414, 2004 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

