Role of Central Amygdala Neuronal Ensembles in Incubation of Nicotine Craving
- PMID: 27535909
- PMCID: PMC4987435
- DOI: 10.1523/JNEUROSCI.1505-16.2016
Role of Central Amygdala Neuronal Ensembles in Incubation of Nicotine Craving
Abstract
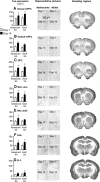
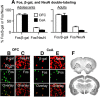
The craving response to smoking-associated cues in humans or to intravenous nicotine-associated cues in adult rats progressively increases or incubates after withdrawal. Here, we further characterized incubation of nicotine craving in the rat model by determining whether this incubation is observed after adolescent-onset nicotine self-administration. We also used the neuronal activity marker Fos and the Daun02 chemogenetic inactivation procedure to identify cue-activated neuronal ensembles that mediate incubation of nicotine craving. We trained adolescent and adult male rats to self-administer nicotine (2 h/d for 12 d) and assessed cue-induced nicotine seeking in extinction tests (1 h) after 1, 7, 14, or 28 withdrawal days. In both adult and adolescent rats, nicotine seeking in the relapse tests followed an inverted U-shaped curve, with maximal responding on withdrawal day 14. Independent of the withdrawal day, nicotine seeking in the relapse tests was higher in adult than in adolescent rats. Analysis of Fos expression in different brain areas of adolescent and adult rats on withdrawal days 1 and 14 showed time-dependent increases in the number of Fos-positive neurons in central and basolateral amygdala, orbitofrontal cortex, ventral and dorsal medial prefrontal cortex, and nucleus accumbens core and shell. In adult Fos-lacZ transgenic rats, selective inactivation of nicotine-cue-activated Fos neurons in central amygdala, but not orbitofrontal cortex, decreased "incubated" nicotine seeking on withdrawal day 14. Our results demonstrate that incubation of nicotine craving occurs after adolescent-onset nicotine self-administration and that neuronal ensembles in central amygdala play a critical role in this incubation.
Significance statement: The craving response to smoking-associated cues in humans or to intravenous nicotine-associated cues in adult rats progressively increases or incubates after withdrawal. It is currently unknown whether incubation of craving also occurs after adolescent-onset nicotine self-administration. The brain areas that mediate such incubation are also unknown. Here, we used a rat model of incubation of drug craving, the neuronal activity marker Fos, and the Daun02 chemogenetic inactivation method to demonstrate that incubation of nicotine craving is also observed after adolescent-onset nicotine self-administration and that neuronal ensembles in the central nucleus of the amygdala play a critical role in this incubation in adult rats.
Keywords: Daun02; Fos; central amygdala; incubation of craving; nicotine; orbitofrontal cortex.
Copyright © 2016 the authors 0270-6474/16/368612-12$15.00/0.
Figures



Similar articles
-
Role of orbitofrontal cortex neuronal ensembles in the expression of incubation of heroin craving.J Neurosci. 2012 Aug 22;32(34):11600-9. doi: 10.1523/JNEUROSCI.1914-12.2012. J Neurosci. 2012. PMID: 22915104 Free PMC article.
-
Selective Inhibition of Amygdala Neuronal Ensembles Encoding Nicotine-Associated Memories Inhibits Nicotine Preference and Relapse.Biol Psychiatry. 2017 Dec 1;82(11):781-793. doi: 10.1016/j.biopsych.2017.04.017. Epub 2017 May 11. Biol Psychiatry. 2017. PMID: 28648649 Free PMC article.
-
Role of Dorsomedial Striatum Neuronal Ensembles in Incubation of Methamphetamine Craving after Voluntary Abstinence.J Neurosci. 2017 Jan 25;37(4):1014-1027. doi: 10.1523/JNEUROSCI.3091-16.2016. J Neurosci. 2017. PMID: 28123032 Free PMC article.
-
Using c-fos to study neuronal ensembles in corticostriatal circuitry of addiction.Brain Res. 2015 Dec 2;1628(Pt A):157-73. doi: 10.1016/j.brainres.2014.11.005. Epub 2014 Nov 11. Brain Res. 2015. PMID: 25446457 Free PMC article. Review.
-
Recent updates on incubation of drug craving: a mini-review.Addict Biol. 2015 Sep;20(5):872-6. doi: 10.1111/adb.12205. Epub 2014 Dec 1. Addict Biol. 2015. PMID: 25440081 Free PMC article. Review.
Cited by
-
Sex differences in the neuroadaptations associated with incubated cocaine-craving: A focus on the dorsomedial prefrontal cortex.Front Behav Neurosci. 2023 Jan 5;16:1027310. doi: 10.3389/fnbeh.2022.1027310. eCollection 2022. Front Behav Neurosci. 2023. PMID: 36688133 Free PMC article.
-
Chasing the addicted engram: identifying functional alterations in Fos-expressing neuronal ensembles that mediate drug-related learned behavior.Learn Mem. 2018 Aug 16;25(9):455-460. doi: 10.1101/lm.046698.117. Print 2018 Sep. Learn Mem. 2018. PMID: 30115767 Free PMC article. Review.
-
Corticostriatal plasticity, neuronal ensembles, and regulation of drug-seeking behavior.Prog Brain Res. 2017;235:93-112. doi: 10.1016/bs.pbr.2017.07.013. Epub 2017 Oct 12. Prog Brain Res. 2017. PMID: 29054293 Free PMC article. Review.
-
Cue-induced nicotine-seeking behavior after withdrawal with or without extinction in rats.Addict Biol. 2018 Jan;23(1):111-119. doi: 10.1111/adb.12480. Epub 2016 Nov 28. Addict Biol. 2018. PMID: 27894160 Free PMC article.
-
Neurobiology of the incubation of drug craving: An update.Pharmacol Rev. 2025 Mar;77(2):100022. doi: 10.1016/j.pharmr.2024.100022. Epub 2024 Nov 29. Pharmacol Rev. 2025. PMID: 40148031 Review.
References
-
- Abdolahi A, Acosta G, Breslin FJ, Hemby SE, Lynch WJ. Incubation of nicotine seeking is associated with enhanced protein kinase A-regulated signaling of dopamine- and cAMP-regulated phosphoprotein of 32 kDa in the insular cortex. Eur J Neurosci. 2010;31:733–741. doi: 10.1111/j.1460-9568.2010.07114.x. - DOI - PMC - PubMed
-
- Airavaara M, Pickens CL, Stern AL, Wihbey KA, Harvey BK, Bossert JM, Liu QR, Hoffer BJ, Shaham Y. Endogenous GDNF in ventral tegmental area and nucleus accumbens does not play a role in the incubation of heroin craving. Addict Biol. 2011;16:261–272. doi: 10.1111/j.1369-1600.2010.00281.x. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
