Mango leaf extract improves central pathology and cognitive impairment in a type 2 diabetes mouse model
- PMID: 27537110
- PMCID: PMC8029052
- DOI: 10.1111/bpa.12433
Mango leaf extract improves central pathology and cognitive impairment in a type 2 diabetes mouse model
Abstract
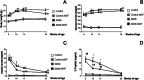
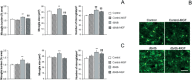
Epidemiological studies reveal that metabolic disorders, and specifically type 2 diabetes (T2D), are relevant risk factors to develop Alzheimer's disease (AD) and vascular dementia (VaD), the most common causes of dementia. AD patients are in a tremendous need of new therapeutic options because of the limited success of available treatments. Natural polyphenols, and concretely Mangifera indica Linn extract (MGF), have been reported to have antiinflammatory, antioxidant and antidiabetic activities. The role of MGF in central complications associated with T2D, after long-term treatment of db/db mice with MGF was analyzed. Metabolic parameters (body weight, glucose and insulin levels) as well as central complications including brain atrophy, inflammatory processes, spontaneous bleeding, tau phosphorylation and cognitive function in db/db mice treated with MGF for 22 weeks were assessed. MGF limits body weight gain in obese db/db mice. Insulin and C-peptide levels, indicative of pancreatic function, were longer maintained in MGF-treated animals. MGF reduced central inflammation by lowering microglia burden, both in the cortex and the hippocampus. Likewise, central spontaneous bleeding was significantly reduced in db/db mice. Cortical and hippocampal atrophy was reduced in db/db mice and tau hyperphosphorylation was lower after MGF treatment, resulting in partial recovery of learning and memory disabilities. Altogether, the data suggested that MGF treatment may provide a useful tool to target different aspects of AD and VaD pathology, and could lead to more effective clinical therapies for the prevention of metabolic related central complications associated with AD and VaD.
Keywords: Alzheimer's disease; diabetes; hemorrhage; inflammation; mango leaf extract; vascular dementia.
© 2016 International Society of Neuropathology.
Figures



Similar articles
-
Long-Term Mangiferin Extract Treatment Improves Central Pathology and Cognitive Deficits in APP/PS1 Mice.Mol Neurobiol. 2017 Aug;54(6):4696-4704. doi: 10.1007/s12035-016-0015-z. Epub 2016 Jul 21. Mol Neurobiol. 2017. PMID: 27443159
-
Central vascular disease and exacerbated pathology in a mixed model of type 2 diabetes and Alzheimer's disease.Psychoneuroendocrinology. 2015 Dec;62:69-79. doi: 10.1016/j.psyneuen.2015.07.606. Epub 2015 Jul 26. Psychoneuroendocrinology. 2015. PMID: 26254770
-
Dementia-like pathology in type-2 diabetes: A novel microRNA mechanism.Mol Cell Neurosci. 2017 Apr;80:58-65. doi: 10.1016/j.mcn.2017.02.005. Epub 2017 Feb 20. Mol Cell Neurosci. 2017. PMID: 28219659 Free PMC article.
-
A look inside the diabetic brain: Contributors to diabetes-induced brain aging.Biochim Biophys Acta. 2009 May;1792(5):444-53. doi: 10.1016/j.bbadis.2008.10.013. Epub 2008 Nov 5. Biochim Biophys Acta. 2009. PMID: 19022375 Free PMC article. Review.
-
Physiological Mechanisms Inherent to Diabetes Involved in the Development of Dementia: Alzheimer's Disease.Neurol Int. 2023 Oct 10;15(4):1253-1272. doi: 10.3390/neurolint15040079. Neurol Int. 2023. PMID: 37873836 Free PMC article. Review.
Cited by
-
Caffeine Restores Neuronal Damage and Inflammatory Response in a Model of Intraventricular Hemorrhage of the Preterm Newborn.Front Cell Dev Biol. 2022 Aug 12;10:908045. doi: 10.3389/fcell.2022.908045. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36035990 Free PMC article.
-
The effects of mango leaf extract during adolescence and adulthood in a rat model of schizophrenia.Front Pharmacol. 2022 Jul 26;13:886514. doi: 10.3389/fphar.2022.886514. eCollection 2022. Front Pharmacol. 2022. PMID: 35959428 Free PMC article.
-
Neuroprotective Effects of Agri-Food By-Products Rich in Phenolic Compounds.Nutrients. 2023 Jan 14;15(2):449. doi: 10.3390/nu15020449. Nutrients. 2023. PMID: 36678322 Free PMC article. Review.
-
Effects of diabetes on microglial physiology: a systematic review of in vitro, preclinical and clinical studies.J Neuroinflammation. 2023 Mar 3;20(1):57. doi: 10.1186/s12974-023-02740-x. J Neuroinflammation. 2023. PMID: 36869375 Free PMC article.
-
Impairment of cognitive flexibility in type 2 diabetic db/db mice.Behav Brain Res. 2019 Oct 3;371:111978. doi: 10.1016/j.bbr.2019.111978. Epub 2019 May 26. Behav Brain Res. 2019. PMID: 31141724 Free PMC article.
References
-
- Biessels GJ, Reijmer YD (2014) Brain changes underlying cognitive dysfunction in diabetes: what can we learn from MRI? Diabetes 63:2244–2252. - PubMed
-
- Biradar SM, Joshi H, Chheda TK (2012) Neuropharmacological effect of Mangiferin on brain cholinesterase and brain biogenic amines in the management of Alzheimer's disease. Eur J Pharmacol 683:140–147. - PubMed
-
- Brundel M, Reijmer YD, van Veluw SJ, Kuijf HJ, Luijten PR, Kappelle LJ, Biessels GJ (2014) Cerebral microvascular lesions on high‐resolution 7‐Tesla MRI in patients with type 2 diabetes. Diabetes 63:3523–3529. - PubMed
-
- Brundel M, van den Heuvel M, de Bresser J, Kappelle LJ, Biessels GJ (2010) Cerebral cortical thickness in patients with type 2 diabetes. J Neurol Sci 299:126–130. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

