Arginase Inhibition Reverses Endothelial Dysfunction, Pulmonary Hypertension, and Vascular Stiffness in Transgenic Sickle Cell Mice
- PMID: 27537757
- PMCID: PMC5032625
- DOI: 10.1213/ANE.0000000000001378
Arginase Inhibition Reverses Endothelial Dysfunction, Pulmonary Hypertension, and Vascular Stiffness in Transgenic Sickle Cell Mice
Abstract
Background: In sickle cell disease (SCD), hemolysis results in the release and activation of arginase, an enzyme that reciprocally regulates nitric oxide (NO) synthase activity and thus, NO production. Simply supplementing the common substrate L-arginine, however, fails to improve NO bioavailability. In this study, we tested the hypothesis that arginase inhibition would improve NO bioavailability and thereby attenuate systemic and pulmonary vascular endothelial dysfunction in transgenic mice with SCD.
Methods: We studied 5-month-old transgenic sickle cell (SC) mice and age matched wild-type (WT) controls. SC mice were treated with the arginase inhibitor, 2(S)-amino-6-boronohexanoic acid (ABH; approximately 400 μg/d) for 4 weeks or left untreated.
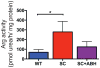
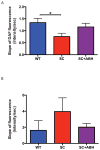
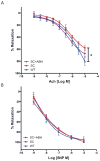
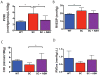
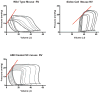
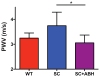
Results: Vascular arginase activity was significantly higher at baseline in untreated SC mice compared to WT controls (SC versus WT, 346 ± 69.3 vs 69 ± 17.3 pmol urea/mg protein/minute; P = 0.0043; n = 4-5 animals per group). Treatment with ABH may significantly decrease arginase activity to levels near WT controls (SC + ABH 125.2 ± 17.3 pmol urea/mg protein/minute; P = 0.0213). Aortic strips from untreated SC mice showed decreased NO and increased reactive oxygen species (ROS) production (NO: fluorescence rate 0.76 ± 0.14 vs 1.34 ± 0.17 RFU/s; P = 0.0005 and ROS: fluorescence rate 3.96 ± 1.70 vs 1.63 ± 1.20 RFU/s, P = 0.0039; n = 3- animals per group). SC animals treated with ABH for 4 weeks demonstrated NO (fluorescence rate: 1.16 ± 0.16) and ROS (fluorescence rate: 2.02 ± 0.45) levels comparable with age-matched WT controls (n = 3- animals per group). The maximal endothelial-dependent vasorelaxation response to acetylcholine was impaired in aortic rings from SC mice compared with WT (57.7% ± 8.4% vs 80.3% ± 11.0%; P = 0.02; n = 6 animals per group). The endothelial-independent response was not different between groups. In SC mice, the right ventricular cardiac output index and end-systolic elastance were similar (4.60 ± 0.51 vs 2.9 ± 0.85 mL/min/100 g and 0.89 ± 0.48 vs 0.58 ± 0.11 mm Hg/μL), whereas the pulmonary vascular resistance index and right ventricular end-systolic pressure were greater (2.9 ± 0.28 vs 5.5 ± 2.0 mm Hg × min/μL/100 g and 18.9 ± 1.1 vs 23.1 ± 4.0 mm Hg; n = 8 animals per group). Pulse wave velocity (a measure of arterial stiffness) was greater in SC mice compared with WT (3.74 ± 0.54 vs 3.25 ± 0.21 m/s; n = 20 animals per group), arginase inhibition for 4 weeks significantly reduced the vascular SC phenotype to one similar to WT animals (P = 0.0009).
Conclusions: Arginase inhibition improves NO bioavailability and thereby attenuates systemic and pulmonary vascular endothelial dysfunction in transgenic mice with SCD. Therefore, arginase is a potential therapeutic target in the treatment of cardiovascular dysfunction in SCD.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Comment in
-
Arginine Therapy Shows Promise for Treatment of Sickle Cell Disease Clinical Subphenotypes of Hemolysis and Arginine Deficiency.Anesth Analg. 2017 Apr;124(4):1369-1370. doi: 10.1213/ANE.0000000000001874. Anesth Analg. 2017. PMID: 28221202 Free PMC article. No abstract available.
Similar articles
-
Obesity-induced vascular dysfunction and arterial stiffening requires endothelial cell arginase 1.Cardiovasc Res. 2017 Nov 1;113(13):1664-1676. doi: 10.1093/cvr/cvx164. Cardiovasc Res. 2017. PMID: 29048462 Free PMC article.
-
Arginase inhibition restores NOS coupling and reverses endothelial dysfunction and vascular stiffness in old rats.J Appl Physiol (1985). 2009 Oct;107(4):1249-57. doi: 10.1152/japplphysiol.91393.2008. Epub 2009 Aug 6. J Appl Physiol (1985). 2009. PMID: 19661445 Free PMC article.
-
Is arginase a potential drug target in tobacco-induced pulmonary endothelial dysfunction?Respir Res. 2015 Mar 28;16(1):46. doi: 10.1186/s12931-015-0196-4. Respir Res. 2015. PMID: 25889611 Free PMC article.
-
Arginase as a potential target in the treatment of cardiovascular disease: reversal of arginine steal?Cardiovasc Res. 2013 Jun 1;98(3):334-43. doi: 10.1093/cvr/cvt036. Epub 2013 Feb 14. Cardiovasc Res. 2013. PMID: 23417041 Review.
-
Arginase and vascular aging.J Appl Physiol (1985). 2008 Nov;105(5):1632-42. doi: 10.1152/japplphysiol.90627.2008. Epub 2008 Aug 21. J Appl Physiol (1985). 2008. PMID: 18719233 Free PMC article. Review.
Cited by
-
Arginase Inhibition Reverses Monocrotaline-Induced Pulmonary Hypertension.Int J Mol Sci. 2017 Jul 25;18(8):1609. doi: 10.3390/ijms18081609. Int J Mol Sci. 2017. PMID: 28757567 Free PMC article.
-
Hydroxyurea improves nitric oxide bioavailability in humanized sickle cell mice.Am J Physiol Regul Integr Comp Physiol. 2021 May 1;320(5):R630-R640. doi: 10.1152/ajpregu.00205.2020. Epub 2021 Feb 24. Am J Physiol Regul Integr Comp Physiol. 2021. PMID: 33624556 Free PMC article.
-
Metabolism in Pulmonary Hypertension.Annu Rev Physiol. 2021 Feb 10;83:551-576. doi: 10.1146/annurev-physiol-031620-123956. Annu Rev Physiol. 2021. PMID: 33566674 Free PMC article. Review.
-
Biochemistry, pharmacology, and in vivo function of arginases.Pharmacol Rev. 2025 Jan;77(1):100015. doi: 10.1124/pharmrev.124.001271. Epub 2024 Nov 22. Pharmacol Rev. 2025. PMID: 39952693 Free PMC article. Review.
-
Cardiovascular Consequences of Sickle Cell Disease.J Blood Med. 2024 May 6;15:207-216. doi: 10.2147/JBM.S455564. eCollection 2024. J Blood Med. 2024. PMID: 38737582 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

