Adverse outcome pathway development from protein alkylation to liver fibrosis
- PMID: 27542122
- PMCID: PMC5364266
- DOI: 10.1007/s00204-016-1814-8
Adverse outcome pathway development from protein alkylation to liver fibrosis
Abstract
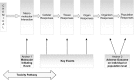
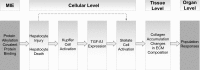
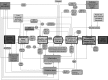
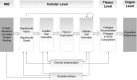
In modern toxicology, substantial efforts are undertaken to develop alternative solutions for in vivo toxicity testing. The adverse outcome pathway (AOP) concept could facilitate knowledge-based safety assessment of chemicals that does not rely exclusively on in vivo toxicity testing. The construction of an AOP is based on understanding toxicological processes at different levels of biological organisation. Here, we present the developed AOP for liver fibrosis and demonstrate a linkage between hepatic injury caused by chemical protein alkylation and the formation of liver fibrosis, supported by coherent and consistent scientific data. This long-term process, in which inflammation, tissue destruction, and repair occur simultaneously, results from the complex interplay between various hepatic cell types, receptors, and signalling pathways. Due to the complexity of the process, an adequate liver fibrosis cell model for in vitro evaluation of a chemical's fibrogenic potential is not yet available. Liver fibrosis poses an important human health issue that is also relevant for regulatory purposes. An AOP described with enough mechanistic detail might support chemical risk assessment by indicating early markers for downstream events and thus facilitating the development of an in vitro testing strategy. With this work, we demonstrate how the AOP framework can support the assembly and coherent display of distributed mechanistic information from the literature to support the use of alternative approaches for prediction of toxicity. This AOP was developed according to the guidance document on developing and assessing AOPs and its supplement, the users' handbook, issued by the Organisation for Economic Co-operation and Development.
Keywords: Adverse outcome pathway (AOP); Alternatives to animal testing; Liver fibrosis; Risk assessment; Systems toxicology.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures
Similar articles
-
Development of an adverse outcome pathway from drug-mediated bile salt export pump inhibition to cholestatic liver injury.Toxicol Sci. 2013 Nov;136(1):97-106. doi: 10.1093/toxsci/kft177. Epub 2013 Aug 14. Toxicol Sci. 2013. PMID: 23945500
-
Development of the adverse outcome pathway "alkylation of DNA in male premeiotic germ cells leading to heritable mutations" using the OECD's users' handbook supplement.Environ Mol Mutagen. 2015 Dec;56(9):724-50. doi: 10.1002/em.21954. Epub 2015 May 22. Environ Mol Mutagen. 2015. PMID: 26010389
-
Implementing Toxicity Testing in the 21st Century (TT21C): Making safety decisions using toxicity pathways, and progress in a prototype risk assessment.Toxicology. 2015 Jun 5;332:102-11. doi: 10.1016/j.tox.2014.02.007. Epub 2014 Feb 25. Toxicology. 2015. PMID: 24582757
-
Adverse outcome pathways as a tool for the design of testing strategies to support the safety assessment of emerging advanced materials at the nanoscale.Part Fibre Toxicol. 2020 May 25;17(1):16. doi: 10.1186/s12989-020-00344-4. Part Fibre Toxicol. 2020. PMID: 32450889 Free PMC article. Review.
-
[Challenge for Adverse Outcome Pathway (AOP)-based Chemical Safety Assessment].Yakugaku Zasshi. 2020;140(4):481-484. doi: 10.1248/yakushi.19-00190-1. Yakugaku Zasshi. 2020. PMID: 32238628 Review. Japanese.
Cited by
-
Ab initio chemical safety assessment: A workflow based on exposure considerations and non-animal methods.Comput Toxicol. 2017 Nov;4:31-44. doi: 10.1016/j.comtox.2017.10.001. Comput Toxicol. 2017. PMID: 29214231 Free PMC article.
-
Mitochondria as the Target of Hepatotoxicity and Drug-Induced Liver Injury: Molecular Mechanisms and Detection Methods.Int J Mol Sci. 2022 Mar 18;23(6):3315. doi: 10.3390/ijms23063315. Int J Mol Sci. 2022. PMID: 35328737 Free PMC article. Review.
-
Natural language processing in toxicology: Delineating adverse outcome pathways and guiding the application of new approach methodologies.Biomater Biosyst. 2022 Jul 28;7:100061. doi: 10.1016/j.bbiosy.2022.100061. eCollection 2022 Aug. Biomater Biosyst. 2022. PMID: 36824484 Free PMC article.
-
Immunomodulatory Effect of Lycium barbarum Polysaccharides against Liver Fibrosis Based on the Intelligent Medical Internet of Things.J Healthc Eng. 2022 Jan 25;2022:6280265. doi: 10.1155/2022/6280265. eCollection 2022. J Healthc Eng. 2022. Retraction in: J Healthc Eng. 2023 Jan 3;2023:9852810. doi: 10.1155/2023/9852810. PMID: 35126934 Free PMC article. Retracted.
-
Stimulation of de novo glutathione synthesis by nitrofurantoin for enhanced resilience of hepatocytes.Cell Biol Toxicol. 2022 Oct;38(5):847-864. doi: 10.1007/s10565-021-09610-3. Epub 2021 May 22. Cell Biol Toxicol. 2022. PMID: 34021431 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

