Targeting prohibitins induces apoptosis in acute myeloid leukemia cells
- PMID: 27542247
- PMCID: PMC5323132
- DOI: 10.18632/oncotarget.11333
Targeting prohibitins induces apoptosis in acute myeloid leukemia cells
Abstract
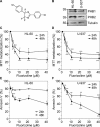
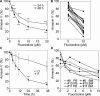
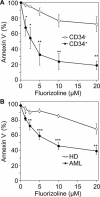
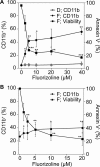
Fluorizoline is a new synthetic molecule that induces apoptosis by selectively targeting prohibitins (PHBs). In this study, the pro-apoptotic effect of fluorizoline was assessed in two cell lines and 21 primary samples from patients with debut of acute myeloid leukemia (AML). Fluorizoline induced apoptosis in AML cells at concentrations in the low micromolar range. All primary samples were sensitive to fluorizoline irrespectively of patients' clinical or genetic features. In addition, fluorizoline inhibited the clonogenic capacity and induced differentiation of AML cells. Fluorizoline increased the mRNA and protein levels of the pro-apoptotic BCL-2 family member NOXA both in cell lines and primary samples analyzed. These results suggest that targeting PHBs could be a new therapeutic strategy for AML.
Keywords: BCL-2 family members; acute myeloid leukemia; apoptosis; cancer; prohibitins.
Conflict of interest statement
D.I.S., S.P., F.A., R.L. and J.G. patented the use of fluorinated thiazolines in the treatment of cancer. The remaining authors declare no conflicts of interest.
Figures


Similar articles
-
The BCL-2 family members NOXA and BIM mediate fluorizoline-induced apoptosis in multiple myeloma cells.Biochem Pharmacol. 2020 Oct;180:114198. doi: 10.1016/j.bcp.2020.114198. Epub 2020 Aug 13. Biochem Pharmacol. 2020. PMID: 32798467
-
A novel prohibitin-binding compound induces the mitochondrial apoptotic pathway through NOXA and BIM upregulation.Oncotarget. 2015 Dec 8;6(39):41750-65. doi: 10.18632/oncotarget.6154. Oncotarget. 2015. PMID: 26497683 Free PMC article.
-
NOXA upregulation by the prohibitin-binding compound fluorizoline is transcriptionally regulated by integrated stress response-induced ATF3 and ATF4.FEBS J. 2021 Feb;288(4):1271-1285. doi: 10.1111/febs.15480. Epub 2020 Jul 26. FEBS J. 2021. PMID: 32648994
-
Mechanistic insights into the antileukemic activity of hyperforin.Curr Cancer Drug Targets. 2013 Jan;13(1):1-10. Curr Cancer Drug Targets. 2013. PMID: 22924417 Review.
-
Targeting the Akt, GSK-3, Bcl-2 axis in acute myeloid leukemia.Adv Biol Regul. 2017 Aug;65:36-58. doi: 10.1016/j.jbior.2017.05.002. Epub 2017 May 19. Adv Biol Regul. 2017. PMID: 28549531 Review.
Cited by
-
The prohibitin-binding compound fluorizoline affects multiple components of the translational machinery and inhibits protein synthesis.J Biol Chem. 2020 Jul 17;295(29):9855-9867. doi: 10.1074/jbc.RA120.012979. Epub 2020 May 19. J Biol Chem. 2020. PMID: 32430400 Free PMC article.
-
Role of Prohibitins in Aging and Therapeutic Potential Against Age-Related Diseases.Front Genet. 2021 Oct 29;12:714228. doi: 10.3389/fgene.2021.714228. eCollection 2021. Front Genet. 2021. PMID: 34868199 Free PMC article. Review.
-
The Prohibitin-Binding Compound Fluorizoline Activates the Integrated Stress Response through the eIF2α Kinase HRI.Int J Mol Sci. 2023 Apr 29;24(9):8064. doi: 10.3390/ijms24098064. Int J Mol Sci. 2023. PMID: 37175767 Free PMC article.
-
Fluorizoline-induced apoptosis requires prohibitins in nematodes and human cells.Apoptosis. 2021 Feb;26(1-2):83-95. doi: 10.1007/s10495-020-01651-z. Epub 2021 Jan 2. Apoptosis. 2021. PMID: 33387147
-
Unravelling Intratumoral Heterogeneity through High-Sensitivity Single-Cell Mutational Analysis and Parallel RNA Sequencing.Mol Cell. 2019 Mar 21;73(6):1292-1305.e8. doi: 10.1016/j.molcel.2019.01.009. Epub 2019 Feb 12. Mol Cell. 2019. PMID: 30765193 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

