How to achieve safe, high-quality clinical studies with non-Medicinal Investigational Products? A practical guideline by using intra-bronchial carbon nanoparticles as case study
- PMID: 27542842
- PMCID: PMC4992213
- DOI: 10.1186/s12931-016-0413-9
How to achieve safe, high-quality clinical studies with non-Medicinal Investigational Products? A practical guideline by using intra-bronchial carbon nanoparticles as case study
Abstract
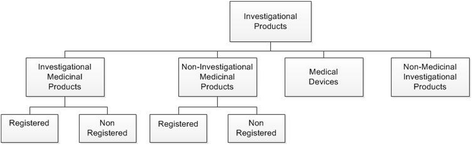
Background: Clinical studies investigating medicinal products need to comply with laws concerning good clinical practice (GCP) and good manufacturing practice (GMP) to guarantee the quality and safety of the product, to protect the health of the participating individual and to assure proper performance of the study. However, there are no specific regulations or guidelines for non-Medicinal Investigational Products (non-MIPs) such as allergens, enriched food supplements, and air pollution components. As a consequence, investigators will avoid clinical research and prefer preclinical models or in vitro testing for e.g. toxicology studies.
The aim of this article is to: 1) briefly review the current guidelines and regulations for Investigational Medicinal Products; 2) present a standardised approach to ensure the quality and safety of non-MIPs in human in vivo research; and 3) discuss some lessons we have learned.
Methods and results: We propose a practical line of approach to compose a clarifying product dossier (PD), comprising the description of the production process, the analysis of the raw and final product, toxicological studies, and a thorough risk-benefit-analysis. This is illustrated by an example from a human in vivo research model to study exposure to air pollutants, by challenging volunteers with a suspension of carbon nanoparticles (the component of ink cartridges for laser printers).
Conclusion: With this novel risk-based approach, the members of competent authorities are provided with standardised information on the quality of the product in relation to the safety of the participants, and the scientific goal of the study.
Keywords: Good clinical practice; Good manufacturing practice; Guidelines; Intervention studies; Legislation; Non-medicinal products.
Figures
Similar articles
-
Typical investigational medicinal products follow relatively uniform regulations in 10 European Clinical Research Infrastructures Network (ECRIN) countries.Trials. 2012 Mar 27;13:27. doi: 10.1186/1745-6215-13-27. Trials. 2012. PMID: 22452964 Free PMC article.
-
The legal framework governing the quality of (traditional) herbal medicinal products in the European Union.J Ethnopharmacol. 2014 Dec 2;158 Pt B:449-53. doi: 10.1016/j.jep.2014.07.044. Epub 2014 Jul 31. J Ethnopharmacol. 2014. PMID: 25086408
-
The need for in situ characterisation in nanosafety assessment: funded transnational access via the QNano research infrastructure.Nanotoxicology. 2013 May;7(3):346-9. doi: 10.3109/17435390.2012.658096. Epub 2012 Feb 14. Nanotoxicology. 2013. PMID: 22280006 Free PMC article. No abstract available.
-
The relevance of pharmacognosy in pharmacological research on herbal medicinal products.Epilepsy Behav. 2015 Nov;52(Pt B):344-62. doi: 10.1016/j.yebeh.2015.05.037. Epub 2015 Jul 10. Epilepsy Behav. 2015. PMID: 26169932 Review.
-
Good Manufacturing Practice (GMP) Compliance for Phage Therapy Medicinal Products.Front Microbiol. 2020 Jun 4;11:1161. doi: 10.3389/fmicb.2020.01161. eCollection 2020. Front Microbiol. 2020. PMID: 32582101 Free PMC article. Review.
Cited by
-
Pulmonary challenge with carbon nanoparticles induces a dose-dependent increase in circulating leukocytes in healthy males.BMC Pulm Med. 2017 Sep 6;17(1):121. doi: 10.1186/s12890-017-0463-x. BMC Pulm Med. 2017. PMID: 28877711 Free PMC article. Clinical Trial.
References
-
- European Commission. Clinical trials-Directive 2001/20/EC of the European Parliament and of the Counsil. In: Official Journal of the European Union. 2001. ec.europa.eu/health/files/eudralex/vol-1/dir_2001_20_en.pdf
-
- Ministry of Health WaS. The Medical Research Involving Human Subjects Act (WMO). 1998. http://wetten.overheid.nl/BWBR0009408
-
- U.S. Food and Drug Administration. Clinical Trials Guidance Documents. 2012. http://www.fda.gov/ScienceResearch/SpecialTopics/RunningClinicalTrials/u....
-
- U.S. Food and Drug Administration. Federal Food, Drug and Cosmetic Act. 1906. http://www.fda.gov/regulatoryinformation/legislation/ucm148690.htm.
-
- World Medical Association. Declaration of Helsinki. Ethical Principles for Medical Research involving Human Subjects. 1964. http://www.wma.net/en/30publications/10polecies/b3/index.html. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials