Micropore-induced capillarity enhances bone distribution in vivo in biphasic calcium phosphate scaffolds
- PMID: 27544807
- PMCID: PMC5045872
- DOI: 10.1016/j.actbio.2016.08.025
Micropore-induced capillarity enhances bone distribution in vivo in biphasic calcium phosphate scaffolds
Abstract
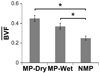
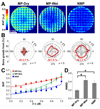
The increasing demand for bone repair solutions calls for the development of efficacious bone scaffolds. Biphasic calcium phosphate (BCP) scaffolds with both macropores and micropores (MP) have improved healing compared to those with macropores and no micropores (NMP), but the role of micropores is unclear. Here, we evaluate capillarity induced by micropores as a mechanism that can affect bone growth in vivo. Three groups of cylindrical scaffolds were implanted in pig mandibles for three weeks: MP were implanted either dry (MP-Dry), or after submersion in phosphate buffered saline, which fills pores with fluid and therefore suppresses micropore-induced capillarity (MP-Wet); NMP were implanted dry. The amount and distribution of bone in the scaffolds were quantified using micro-computed tomography. MP-Dry had a more homogeneous bone distribution than MP-Wet, although the average bone volume fraction, BVF‾, was not significantly different for these two groups (0.45±0.03 and 0.37±0.03, respectively). There was no significant difference in the radial bone distribution of NMP and MP-Wet, but the BVF‾, of NMP was significantly lower among the three groups (0.25±0.02). These results suggest that micropore-induced capillarity enhances bone regeneration by improving the homogeneity of bone distribution in BCP scaffolds. The explicit design and use of capillarity in bone scaffolds may lead to more effective treatments of large and complex bone defects.
Statement of significance: The increasing demand for bone repair calls for more efficacious bone scaffolds and calcium phosphate-based materials are considered suitable for this application. Macropores (>100μm) are necessary for bone ingrowth and vascularization. However, studies have shown that microporosity (<20μm) also enhances growth, but there is no consensus on the controlling mechanisms. In previous in vitro work, we suggested that micropore-induced capillarity had the potential to enhance bone growth in vivo. This work illustrates the positive effects of capillarity on bone regeneration in vivo; it demonstrates that micropore-induced capillarity significantly enhances the bone distribution in the scaffold. The results will impact the design of scaffolds to better exploit capillarity and improve treatments for large and load-bearing bone defects.
Keywords: Bone regeneration; Calcium phosphate scaffold; Capillary action; Micro-computed tomography; Microporosity.
Copyright © 2016 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
Figures




Similar articles
-
The effect of BMP-2 on micro- and macroscale osteointegration of biphasic calcium phosphate scaffolds with multiscale porosity.Acta Biomater. 2010 Aug;6(8):3283-91. doi: 10.1016/j.actbio.2010.02.026. Epub 2010 Feb 20. Acta Biomater. 2010. PMID: 20176148
-
Mineralization in micropores of calcium phosphate scaffolds.Acta Biomater. 2019 Jan 1;83:435-455. doi: 10.1016/j.actbio.2018.11.003. Epub 2018 Nov 5. Acta Biomater. 2019. PMID: 30408560 Review.
-
Analysis of the roles of microporosity and BMP-2 on multiple measures of bone regeneration and healing in calcium phosphate scaffolds.Acta Biomater. 2011 Apr;7(4):1760-71. doi: 10.1016/j.actbio.2010.12.030. Epub 2010 Dec 31. Acta Biomater. 2011. PMID: 21199692
-
Multiscale Porosity Directs Bone Regeneration in Biphasic Calcium Phosphate Scaffolds.ACS Biomater Sci Eng. 2017 Nov 13;3(11):2768-2778. doi: 10.1021/acsbiomaterials.6b00632. Epub 2016 Dec 13. ACS Biomater Sci Eng. 2017. PMID: 33418701
-
Effect of the biodegradation rate controlled by pore structures in magnesium phosphate ceramic scaffolds on bone tissue regeneration in vivo.Acta Biomater. 2016 Oct 15;44:155-67. doi: 10.1016/j.actbio.2016.08.039. Epub 2016 Aug 21. Acta Biomater. 2016. PMID: 27554019
Cited by
-
Evaluation of Osteoconduction of Biphasic Calcium Phosphate Ceramic in the Calvaria of Rats: Microscopic and Histometric Analysis.J Funct Biomater. 2019 Jan 17;10(1):7. doi: 10.3390/jfb10010007. J Funct Biomater. 2019. PMID: 30658427 Free PMC article.
-
Development, Characterization and In Vitro Biological Properties of Scaffolds Fabricated From Calcium Phosphate Nanoparticles.Int J Mol Sci. 2019 Apr 11;20(7):1790. doi: 10.3390/ijms20071790. Int J Mol Sci. 2019. PMID: 30978933 Free PMC article.
-
Advancements in Bone Replacement Techniques-Potential Uses After Maxillary and Mandibular Resections Due to Medication-Related Osteonecrosis of the Jaw (MRONJ).Cells. 2025 Jan 20;14(2):145. doi: 10.3390/cells14020145. Cells. 2025. PMID: 39851573 Free PMC article. Review.
-
3D printed gelatin/decellularized bone composite scaffolds for bone tissue engineering: Fabrication, characterization and cytocompatibility study.Mater Today Bio. 2022 Jun 6;15:100309. doi: 10.1016/j.mtbio.2022.100309. eCollection 2022 Jun. Mater Today Bio. 2022. PMID: 35757025 Free PMC article.
-
Biodegradable composites with antibiotics and growth factors for dual release kinetics.J Mater Sci Mater Med. 2024 Jul 29;35(1):40. doi: 10.1007/s10856-024-06809-8. J Mater Sci Mater Med. 2024. PMID: 39073605 Free PMC article.
References
-
- Kinaci A, Neuhaus V, Ring DC. Trends in bone graft use in the United States. Orthopedics. 2014;37:e783–8. - PubMed
-
- Habraken W, Habibovic P, Epple M, Bohner M. Calcium phosphates in biomedical applications: materials for the future? Mater Today. 2015
-
- UCBPI Office. Census Bureau Releases Comprehensive Analysis of Fast-Growing 90-and-Older Population - Aging Population - Newsroom - U.S. Census Bureau. [accessed March 14, 2016]; (n.d.). https://www.census.gov/newsroom/releases/archives/aging_population/cb11-....
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

