The viral capping enzyme nsP1: a novel target for the inhibition of chikungunya virus infection
- PMID: 27545976
- PMCID: PMC4992889
- DOI: 10.1038/srep31819
The viral capping enzyme nsP1: a novel target for the inhibition of chikungunya virus infection
Abstract
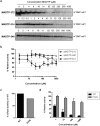
The chikungunya virus (CHIKV) has become a substantial global health threat due to its massive re-emergence, the considerable disease burden and the lack of vaccines or therapeutics. We discovered a novel class of small molecules ([1,2,3]triazolo[4,5-d]pyrimidin-7(6H)-ones) with potent in vitro activity against CHIKV isolates from different geographical regions. Drug-resistant variants were selected and these carried a P34S substitution in non-structural protein 1 (nsP1), the main enzyme involved in alphavirus RNA capping. Biochemical assays using nsP1 of the related Venezuelan equine encephalitis virus revealed that the compounds specifically inhibit the guanylylation of nsP1. This is, to the best of our knowledge, the first report demonstrating that the alphavirus capping machinery is an excellent antiviral drug target. Considering the lack of options to treat CHIKV infections, this series of compounds with their unique (alphavirus-specific) target offers promise for the development of therapy for CHIKV infections.
Figures
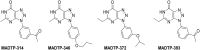



Similar articles
-
Antiviral activity of [1,2,3]triazolo[4,5-d]pyrimidin-7(6H)-ones against chikungunya virus targeting the viral capping nsP1.Antiviral Res. 2017 Aug;144:216-222. doi: 10.1016/j.antiviral.2017.06.003. Epub 2017 Jun 12. Antiviral Res. 2017. PMID: 28619679
-
6'-β-Fluoro-Homoaristeromycin and 6'-Fluoro-Homoneplanocin A Are Potent Inhibitors of Chikungunya Virus Replication through Their Direct Effect on Viral Nonstructural Protein 1.Antimicrob Agents Chemother. 2020 Mar 24;64(4):e02532-19. doi: 10.1128/AAC.02532-19. Print 2020 Mar 24. Antimicrob Agents Chemother. 2020. PMID: 31964798 Free PMC article.
-
Identification of small molecule inhibitors of the Chikungunya virus nsP1 RNA capping enzyme.Antiviral Res. 2018 Jun;154:124-131. doi: 10.1016/j.antiviral.2018.03.013. Epub 2018 Apr 20. Antiviral Res. 2018. PMID: 29680670 Free PMC article.
-
Advances in the Development of Non-Structural Protein 1 (NsP1) Inhibitors for the Treatment of Chikungunya Virus Infection.Mini Rev Med Chem. 2024;24(22):1972-1982. doi: 10.2174/0113895575301735240607055839. Mini Rev Med Chem. 2024. PMID: 38910486 Review.
-
Chikungunya virus: an update on antiviral development and challenges.Drug Discov Today. 2013 Oct;18(19-20):969-83. doi: 10.1016/j.drudis.2013.05.002. Epub 2013 May 15. Drug Discov Today. 2013. PMID: 23684571 Free PMC article. Review.
Cited by
-
Antivirals against the Chikungunya Virus.Viruses. 2021 Jul 5;13(7):1307. doi: 10.3390/v13071307. Viruses. 2021. PMID: 34372513 Free PMC article. Review.
-
A compendium of small molecule direct-acting and host-targeting inhibitors as therapies against alphaviruses.J Antimicrob Chemother. 2017 Nov 1;72(11):2973-2989. doi: 10.1093/jac/dkx224. J Antimicrob Chemother. 2017. PMID: 28981632 Free PMC article. Review.
-
Identification and evaluation of potential SARS-CoV-2 antiviral agents targeting mRNA cap guanine N7-Methyltransferase.Antiviral Res. 2021 Sep;193:105142. doi: 10.1016/j.antiviral.2021.105142. Epub 2021 Jul 23. Antiviral Res. 2021. PMID: 34303749 Free PMC article.
-
Advances in the Development of Small Molecule Antivirals against Equine Encephalitic Viruses.Viruses. 2023 Feb 1;15(2):413. doi: 10.3390/v15020413. Viruses. 2023. PMID: 36851628 Free PMC article. Review.
-
The RNA Capping Enzyme Domain in Protein A is Essential for Flock House Virus Replication.Viruses. 2018 Sep 9;10(9):483. doi: 10.3390/v10090483. Viruses. 2018. PMID: 30205593 Free PMC article.
References
-
- Chen L. H. & Wilson M. E. Dengue and chikungunya in travelers. Current Opinion in Infectious Diseases 25, 523–529 (2012). - PubMed
-
- Leparc-Goffart I., Nougairede A., Cassadou S., Prat C. & De Lamballerie X. Chikungunya in the Americas. The Lancet 383, 514 (2014). - PubMed
-
- Hoz J. M. D. La et al.. Fatal cases of Chikungunya virus infection in Colombia: Diagnostic and treatment challenges. J. Clin. Virol. 69, 27–29 (2015). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

