Transcriptional Reactivation of the FMR1 Gene. A Possible Approach to the Treatment of the Fragile X Syndrome
- PMID: 27548224
- PMCID: PMC4999837
- DOI: 10.3390/genes7080049
Transcriptional Reactivation of the FMR1 Gene. A Possible Approach to the Treatment of the Fragile X Syndrome
Abstract
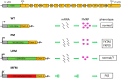
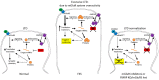
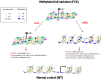
Fragile X syndrome (FXS) is the most common cause of inherited intellectual disability, caused by CGG expansion over 200 repeats (full mutation, FM) at the 5' untranslated region (UTR) of the fragile X mental retardation 1 (FMR1) gene and subsequent DNA methylation of the promoter region, accompanied by additional epigenetic histone modifications that result in a block of transcription and absence of the fragile X mental retardation protein (FMRP). The lack of FMRP, involved in multiple aspects of mRNA metabolism in the brain, is thought to be the direct cause of the FXS phenotype. Restoration of FMR1 transcription and FMRP production can be obtained in vitro by treating FXS lymphoblastoid cell lines with the demethylating agent 5-azadeoxycytidine, demonstrating that DNA methylation is key to FMR1 inactivation. This concept is strengthened by the existence of rare male carriers of a FM, who are unable to methylate the FMR1 promoter. These individuals produce limited amounts of FMRP and are of normal intelligence. Their inability to methylate the FMR1 promoter, whose cause is not yet fully elucidated, rescues them from manifesting the FXS. These observations demonstrate that a therapeutic approach to FXS based on the pharmacological reactivation of the FMR1 gene is conceptually tenable and worthy of being further pursued.
Keywords: DNA methylation; FMR1 gene; Fragile X syndrome; drug treatments; epigenetic therapy; histone modifications.
Figures

Similar articles
-
Epigenetic characterization of the FMR1 gene and aberrant neurodevelopment in human induced pluripotent stem cell models of fragile X syndrome.PLoS One. 2011;6(10):e26203. doi: 10.1371/journal.pone.0026203. Epub 2011 Oct 12. PLoS One. 2011. PMID: 22022567 Free PMC article.
-
DNA Methylation, Mechanisms of FMR1 Inactivation and Therapeutic Perspectives for Fragile X Syndrome.Biomolecules. 2021 Feb 16;11(2):296. doi: 10.3390/biom11020296. Biomolecules. 2021. PMID: 33669384 Free PMC article. Review.
-
The feasibility and utility of hair follicle sampling to measure FMRP and FMR1 mRNA in children with or without fragile X syndrome: a pilot study.J Neurodev Disord. 2022 Dec 9;14(1):57. doi: 10.1186/s11689-022-09465-7. J Neurodev Disord. 2022. PMID: 36494616 Free PMC article.
-
Transcriptomic profiling of unmethylated full mutation carriers implicates TET3 in FMR1 CGG repeat expansion methylation dynamics in fragile X syndrome.J Neurodev Disord. 2025 Apr 26;17(1):22. doi: 10.1186/s11689-025-09609-5. J Neurodev Disord. 2025. PMID: 40287634 Free PMC article.
-
Pharmacological Reactivation of the Silenced FMR1 Gene as a Targeted Therapeutic Approach for Fragile X Syndrome.Brain Sci. 2019 Feb 12;9(2):39. doi: 10.3390/brainsci9020039. Brain Sci. 2019. PMID: 30759772 Free PMC article. Review.
Cited by
-
Rescue of Fragile X Syndrome Neurons by DNA Methylation Editing of the FMR1 Gene.Cell. 2018 Feb 22;172(5):979-992.e6. doi: 10.1016/j.cell.2018.01.012. Epub 2018 Feb 15. Cell. 2018. PMID: 29456084 Free PMC article.
-
Prenatal Diagnosis of Fragile X: Can a Full Mutation Allele in the FMR1 Gene Contract to a Normal Size?Front Genet. 2017 Nov 3;8:158. doi: 10.3389/fgene.2017.00158. eCollection 2017. Front Genet. 2017. PMID: 29163631 Free PMC article.
-
Epigenetic insights into Fragile X Syndrome.Front Cell Dev Biol. 2024 Aug 16;12:1432444. doi: 10.3389/fcell.2024.1432444. eCollection 2024. Front Cell Dev Biol. 2024. PMID: 39220684 Free PMC article. Review.
-
Fragile X-related protein family: a double-edged sword in neurodevelopmental disorders and cancer.Crit Rev Biochem Mol Biol. 2020 Oct;55(5):409-424. doi: 10.1080/10409238.2020.1810621. Epub 2020 Sep 2. Crit Rev Biochem Mol Biol. 2020. PMID: 32878499 Free PMC article. Review.
-
Association between IQ and FMR1 protein (FMRP) across the spectrum of CGG repeat expansions.PLoS One. 2019 Dec 31;14(12):e0226811. doi: 10.1371/journal.pone.0226811. eCollection 2019. PLoS One. 2019. PMID: 31891607 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

