Lesion of the rostromedial tegmental nucleus increases voluntary ethanol consumption and accelerates extinction of ethanol-induced conditioned taste aversion
- PMID: 27549757
- PMCID: PMC5063894
- DOI: 10.1007/s00213-016-4406-7
Lesion of the rostromedial tegmental nucleus increases voluntary ethanol consumption and accelerates extinction of ethanol-induced conditioned taste aversion
Abstract
Rationale: Ethanol has rewarding and aversive properties, and the balance of these properties influences voluntary ethanol consumption. Preclinical and clinical evidence show that the aversive properties of ethanol limit intake. The neural circuits underlying ethanol-induced aversion learning are not fully understood. We have previously shown that the lateral habenula (LHb), a region critical for aversive conditioning, plays an important role in ethanol-directed behaviors. However, the neurocircuitry through which LHb exerts its actions is unknown.
Objective: In the present study, we investigate a role for the rostromedial tegmental nucleus (RMTg), a major LHb projection target, in regulating ethanol-directed behaviors.
Methods: Rats received either sham or RMTg lesions and were studied during voluntary ethanol consumption; operant ethanol self-administration, extinction, and yohimbine-induced reinstatement of ethanol-seeking; and ethanol-induced conditioned taste aversion (CTA).
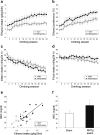
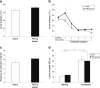
Results: RMTg lesions increased voluntary ethanol consumption and accelerated extinction of ethanol-induced CTA.
Conclusions: The RMTg plays an important role in regulating voluntary ethanol consumption, possibly by mediating ethanol-induced aversive conditioning.
Keywords: Aversion; Ethanol; Lateral habenula (LHb); Reward; Rostromedial tegmental nucleus (RMTg).
Conflict of interest statement
Compliance with ethical standards All procedures used were approved by the University of Utah Animal Care and Use Committee and carried out in accordance with the Guide for the Care and Use of Laboratory Animals (eighth edition). Funding Funding support was provided by the National Institutes Health under award MH094870. Conflict of interest The authors declare that they have no conflict of interest.
Figures



Similar articles
-
Role for the Rostromedial Tegmental Nucleus in Signaling the Aversive Properties of Alcohol.Alcohol Clin Exp Res. 2016 Aug;40(8):1651-61. doi: 10.1111/acer.13140. Epub 2016 Jul 8. Alcohol Clin Exp Res. 2016. PMID: 27388762 Free PMC article.
-
Lesions of the lateral habenula increase voluntary ethanol consumption and operant self-administration, block yohimbine-induced reinstatement of ethanol seeking, and attenuate ethanol-induced conditioned taste aversion.PLoS One. 2014 Apr 2;9(4):e92701. doi: 10.1371/journal.pone.0092701. eCollection 2014. PLoS One. 2014. PMID: 24695107 Free PMC article.
-
Excitation of lateral habenula neurons as a neural mechanism underlying ethanol-induced conditioned taste aversion.J Physiol. 2017 Feb 15;595(4):1393-1412. doi: 10.1113/JP272994. Epub 2016 Nov 8. J Physiol. 2017. PMID: 27682823 Free PMC article.
-
Decoding the dual regulatory systems: Contemporary insights into the lateral habenula and rostromedial tegmental nucleus physiology.Neurobiol Dis. 2025 Aug 5;214:107049. doi: 10.1016/j.nbd.2025.107049. Online ahead of print. Neurobiol Dis. 2025. PMID: 40769282 Review.
-
Ethanol drinking in rodents: is free-choice drinking related to the reinforcing effects of ethanol?Alcohol. 2008 Feb;42(1):1-11. doi: 10.1016/j.alcohol.2007.10.005. Alcohol. 2008. PMID: 18164576 Free PMC article. Review.
Cited by
-
Rostromedial tegmental nucleus nociceptin/orphanin FQ (N/OFQ) signaling regulates anxiety- and depression-like behaviors in alcohol withdrawn rats.Neuropsychopharmacology. 2023 May;48(6):908-919. doi: 10.1038/s41386-022-01482-3. Epub 2022 Nov 3. Neuropsychopharmacology. 2023. PMID: 36329156 Free PMC article.
-
The rostromedial tegmental (RMTg) "brake" on dopamine and behavior: A decade of progress but also much unfinished work.Neuropharmacology. 2021 Oct 15;198:108763. doi: 10.1016/j.neuropharm.2021.108763. Epub 2021 Aug 22. Neuropharmacology. 2021. PMID: 34433088 Free PMC article. Review.
-
Yohimbine as a pharmacological probe for alcohol research: a systematic review of rodent and human studies.Neuropsychopharmacology. 2022 Nov;47(12):2111-2122. doi: 10.1038/s41386-022-01363-9. Epub 2022 Jun 27. Neuropsychopharmacology. 2022. PMID: 35760866 Free PMC article.
-
The Establishment of a CSF-Contacting Nucleus "Knockout" Model Animal.Front Neuroanat. 2018 Mar 27;12:22. doi: 10.3389/fnana.2018.00022. eCollection 2018. Front Neuroanat. 2018. PMID: 29636668 Free PMC article.
-
Low-dose ethanol excites lateral habenula neurons projecting to VTA, RMTg, and raphe.Int J Physiol Pathophysiol Pharmacol. 2017 Dec 25;9(6):217-230. eCollection 2017. Int J Physiol Pathophysiol Pharmacol. 2017. PMID: 29348799 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

