c-MYC responds to glucose deprivation in a cell-type-dependent manner
- PMID: 27551483
- PMCID: PMC4979460
- DOI: 10.1038/cddiscovery.2015.57
c-MYC responds to glucose deprivation in a cell-type-dependent manner
Abstract
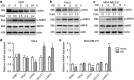
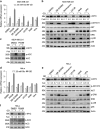
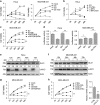
Metabolic reprogramming supports cancer cells' demands for rapid proliferation and growth. Previous work shows that oncogenes, such as MYC, hypoxia-inducible factor 1 (HIF1), have a central role in driving metabolic reprogramming. A lot of metabolic enzymes, which are deregulated in most cancer cells, are the targets of these oncogenes. However, whether metabolic change affects these oncogenes is still unclear. Here we show that glucose deprivation (GD) affects c-MYC protein levels in a cell-type-dependent manner regardless of P53 mutation status. GD dephosphorylates and then decreases c-MYC protein stability through PI3K signaling pathway in HeLa cells, but not in MDA-MB-231 cells. Role of c-MYC in sensitivity of GD also varies with cell types. c-MYC-mediated glutamine metabolism partially improves the sensitivity of GD in MDA-MB-231 cells. Our results reveal that the heterogeneity of cancer cells in response to metabolic stress should be considered in metabolic therapy for cancer.
Figures



Similar articles
-
Metabolic Roles of HIF1, c-Myc, and p53 in Glioma Cells.Metabolites. 2024 Apr 25;14(5):249. doi: 10.3390/metabo14050249. Metabolites. 2024. PMID: 38786726 Free PMC article. Review.
-
Inhibitor of differentiation 1 transcription factor promotes metabolic reprogramming in hepatocellular carcinoma cells.FASEB J. 2016 Jan;30(1):262-75. doi: 10.1096/fj.15-277749. Epub 2015 Sep 1. FASEB J. 2016. PMID: 26330493 Free PMC article.
-
Oncogenic regulation of tumor metabolic reprogramming.Oncotarget. 2016 Sep 20;7(38):62726-62753. doi: 10.18632/oncotarget.10911. Oncotarget. 2016. PMID: 28040803 Free PMC article. Review.
-
Causes and Consequences of A Glutamine Induced Normoxic HIF1 Activity for the Tumor Metabolism.Int J Mol Sci. 2019 Sep 24;20(19):4742. doi: 10.3390/ijms20194742. Int J Mol Sci. 2019. PMID: 31554283 Free PMC article.
-
MYC Expression and Metabolic Redox Changes in Cancer Cells: A Synergy Able to Induce Chemoresistance.Oxid Med Cell Longev. 2019 Jun 25;2019:7346492. doi: 10.1155/2019/7346492. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31341534 Free PMC article. Review.
Cited by
-
T Cell Metabolism in Infection.Front Immunol. 2022 Mar 14;13:840610. doi: 10.3389/fimmu.2022.840610. eCollection 2022. Front Immunol. 2022. PMID: 35359994 Free PMC article. Review.
-
Intervenolin suppresses gastric cancer cell growth through the induction of TSP-1 secretion from fibroblast-like stromal cells.Oncol Lett. 2018 Nov;16(5):6777-6785. doi: 10.3892/ol.2018.9485. Epub 2018 Sep 21. Oncol Lett. 2018. PMID: 30405822 Free PMC article.
-
Succinate dehydrogenase B-deficient cancer cells are highly sensitive to bromodomain and extra-terminal inhibitors.Oncotarget. 2017 Apr 25;8(17):28922-28938. doi: 10.18632/oncotarget.15959. Oncotarget. 2017. PMID: 28423651 Free PMC article.
-
LncRNA-MIF, a c-Myc-activated long non-coding RNA, suppresses glycolysis by promoting Fbxw7-mediated c-Myc degradation.EMBO Rep. 2016 Aug;17(8):1204-20. doi: 10.15252/embr.201642067. Epub 2016 Jun 17. EMBO Rep. 2016. PMID: 27317567 Free PMC article.
-
LncRNA FTO-IT1 promotes glycolysis and progression of hepatocellular carcinoma through modulating FTO-mediated N6-methyladenosine modification on GLUT1 and PKM2.J Exp Clin Cancer Res. 2023 Oct 16;42(1):267. doi: 10.1186/s13046-023-02847-2. J Exp Clin Cancer Res. 2023. PMID: 37840133 Free PMC article.
References
-
- Warburg O . On respiratory impairment in cancer cells. Science 1956; 124: 269–270. - PubMed
-
- Koppenol WH , Bounds PL , Dang CV . Otto Warburg's contributions to current concepts of cancer metabolism. Nat Rev Cancer 2011; 11: 325–337. - PubMed
-
- Postovit LM , Adams MA , Lash GE , Heaton JP , Graham CH . Oxygen-mediated regulation of tumor cell invasiveness. Involvement of a nitric oxide signaling pathway. J Biol Chem 2002; 277: 35730–35737. - PubMed
-
- Krtolica A , Ludlow JW . Hypoxia arrests ovarian carcinoma cell cycle progression, but invasion is unaffected. Cancer Res 1996; 56: 1168–1173. - PubMed
-
- Yamamoto T , Seino Y , Fukumoto H , Koh G , Yano H , Inagaki N et al. Over-expression of facilitative glucose transporter genes in human cancer. Biochem Biophys Res Commun 1990; 170: 223–230. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

