Selective cytoprotective effect of histamine on doxorubicin-induced hepatic and cardiac toxicity in animal models
- PMID: 27551485
- PMCID: PMC4979467
- DOI: 10.1038/cddiscovery.2015.59
Selective cytoprotective effect of histamine on doxorubicin-induced hepatic and cardiac toxicity in animal models
Abstract
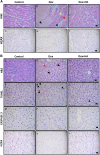
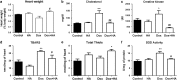
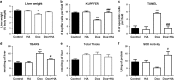
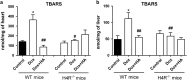
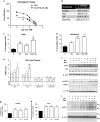
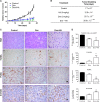
The aim of the present work was to evaluate the potential protective effect of histamine on Doxorubicin (Dox)-induced hepatic and cardiac toxicity in different rodent species and in a triple-negative breast tumor-bearing mice model. Male Sprague Dawley rats and Balb/c mice were divided into four groups: control (received saline), histamine (5 mg/kg for rats and 1 mg/kg for mice, daily subcutaneous injection starting 24 h before treatment with Dox), Dox (2 mg/kg, intraperitoneally injected three times a week for 2 weeks) and Dox+histamine (received both treatments). Tissue toxicity was evaluated by histopathological studies and oxidative stress and biochemical parameters. The combined effect of histamine and Dox was also investigated in vitro and in vivo in human MDA-MB-231 triple-negative breast cancer model. Heart and liver of Dox-treated animals displayed severe histological damage, loss of tissue weight, increased TBARS levels and DNA damage along with an augment in serum creatine kinase-myocardial band. Pretreatment with histamine prevented Dox-induced tissue events producing a significant preservation of the integrity of both rat and mouse myocardium and liver, through the reduction of Dox-induced oxidative stress and apoptosis. Histamine treatment preserved anti-tumor activity of Dox, exhibiting differential cytotoxicity and increasing the Dox-induced inhibition of breast tumor growth. Findings provide preclinical evidence indicating that histamine could be a promising candidate as a selective cytoprotective agent for the treatment of Dox-induced cardiac and hepatic toxicity, and encourage the translation to clinical practice.
Figures


Similar articles
-
Crocin protects against doxorubicin-induced myocardial toxicity in rats through down-regulation of inflammatory and apoptic pathways.Chem Biol Interact. 2016 Mar 5;247:39-48. doi: 10.1016/j.cbi.2016.01.014. Epub 2016 Jan 22. Chem Biol Interact. 2016. PMID: 26807765
-
Allicin ameliorates doxorubicin-induced cardiotoxicity in rats via suppression of oxidative stress, inflammation and apoptosis.Cancer Chemother Pharmacol. 2017 Oct;80(4):745-753. doi: 10.1007/s00280-017-3413-7. Epub 2017 Aug 7. Cancer Chemother Pharmacol. 2017. PMID: 28785995
-
α-Linolenic acid attenuates doxorubicin-induced cardiotoxicity in rats through suppression of oxidative stress and apoptosis.Acta Biochim Biophys Sin (Shanghai). 2013 Oct;45(10):817-26. doi: 10.1093/abbs/gmt082. Epub 2013 Jul 28. Acta Biochim Biophys Sin (Shanghai). 2013. PMID: 23896563
-
Ameliorative Effect of Cardamom Aqueous Extract on Doxorubicin-Induced Cardiotoxicity in Rats.Cells Tissues Organs. 2018;206(1-2):62-72. doi: 10.1159/000496109. Epub 2019 Feb 4. Cells Tissues Organs. 2018. PMID: 30716735
-
Protective effects of tannic acid on acute doxorubicin-induced cardiotoxicity: Involvement of suppression in oxidative stress, inflammation, and apoptosis.Biomed Pharmacother. 2017 Sep;93:1253-1260. doi: 10.1016/j.biopha.2017.07.051. Epub 2017 Jul 20. Biomed Pharmacother. 2017. PMID: 28738542
Cited by
-
Histamine H4 Receptor Agonism Induces Antitumor Effects in Human T-Cell Lymphoma.Int J Mol Sci. 2022 Jan 26;23(3):1378. doi: 10.3390/ijms23031378. Int J Mol Sci. 2022. PMID: 35163302 Free PMC article.
-
Histamine receptors and cancer pharmacology: an update.Br J Pharmacol. 2020 Feb;177(3):516-538. doi: 10.1111/bph.14535. Epub 2018 Dec 13. Br J Pharmacol. 2020. PMID: 30414378 Free PMC article. Review.
-
Doxorubicin Paradoxically Ameliorates Tumor-Induced Inflammation in Young Mice.Int J Mol Sci. 2021 Aug 21;22(16):9023. doi: 10.3390/ijms22169023. Int J Mol Sci. 2021. PMID: 34445729 Free PMC article.
-
Histamine: a potential cytoprotective agent to improve cancer therapy?Cell Death Dis. 2015 Dec 31;6(12):e2029. doi: 10.1038/cddis.2015.378. Cell Death Dis. 2015. PMID: 26720338 Free PMC article. No abstract available.
-
Pristimerin protects against doxorubicin-induced cardiotoxicity and fibrosis through modulation of Nrf2 and MAPK/NF-kB signaling pathways.Cancer Manag Res. 2018 Dec 19;11:47-61. doi: 10.2147/CMAR.S186696. eCollection 2019. Cancer Manag Res. 2018. PMID: 30588110 Free PMC article.
References
-
- Ferlay J , Soerjomataram I , Dikshit R , Eser S , Mathers C , Rebelo M et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015; 136: E359–E386. - PubMed
-
- Octavia Y , Tocchetti CG , Gabrielson KL , Janssens S , Crijns HJ , Moens AL . Doxorubicin-induced cardiomyopathy: from molecular mechanisms to therapeutic strategies. J Mol Cell Cardiol 2012; 52: 1213–1225. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

