Total saponins of panaxnotoginseng promotes lymphangiogenesis by activation VEGF-C expression of lymphatic endothelial cells
- PMID: 27553977
- PMCID: PMC5108701
- DOI: 10.1016/j.jep.2016.08.032
Total saponins of panaxnotoginseng promotes lymphangiogenesis by activation VEGF-C expression of lymphatic endothelial cells
Abstract
Ethnopharmacological relevance: Lymphatic system plays an important role in maintaining the fluid homeostasis and normal immune responses, anatomic or functional obstruction of which leads to lymphedema, and treatments for therapeutic lymphangiogenesis are efficiency for secondary lymphedema. Total saponins of panaxnotoginseng (PNS) are a mixture isolated from Panaxnotoginseng (Burkill) F.H.Chen, which has been used as traditional Chinese medicine in China for treatment of cardio- and cerebro-vascular diseases. The aim of this study was to determine the effect and mechanism of PNS on lymphangiogenesis.
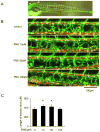
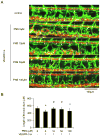
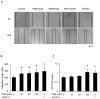
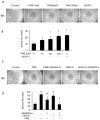
Methods: The Tg (fli1: egfp; gata1: dsred) transgenic zebrafish embryos were treated with different concentrations of PNS (10, 50, 100μM) for 48h with or without the 6h pretreatment of the 30μM Vascular endothelial growth factors receptor (VEGFR)-3 kinase inhibitor, followed with morphological observation and lympangiogenesis of thoracic duct assessment. The effect of PNS on cell viability, migration, tube formation and Vascular endothelial growth factors (VEGF)-C mRNA and protein expression of lymphatic endothelial cells (LECs) were determined. The role of phosphatidylinositol-3 (PI-3)-kinase (PI3K), extracellular signal-regulated kinase (ERK)1/2 pathways, c-Jun N-terminal kinase (JNK) and P38 mitogen activated protein kinases (MAPK) signaling in PNS-induced VEGF-C expression of LECs by using pharmacological agents to block each signal.
Results: PNS promotes lymphangiogenesis of thoracic duct in zebrafish with or without VEGFR3 Kinase inhibitor pre-impairment. PNS promotes proliferation, migration and tube formation of LECs. The tube formation induced by PNS could be blocked by VEGFR3 Kinase inhibitor. PNS induce VEGF-C expression of LEC, which could be blocked by ERK1/2, PI3K and P38MAPK signaling inhibitors.
Conclusion: PNS activates lymphangiogenesis both in vivo and in vitro by up-regulating VEGF-C expression and activation of ERK1/2, PI3K and P38MAPK signaling. These findings provide a novel insight into the role of PNS in lymphangiogenesis and suggest that it might be an attractive and suitable therapeutic agent for treating secondary lymphedema or other lymphatic system impairment related disease.
Keywords: Acrylamide (PubChemCID: 6579); Ammonium Persulfate (PubChemCID:62648); Chloroform (PubChemCID:6212); Dimethyl sulfoxide (DMSO) (PubChem CID: 679); Ethanol (PubChem CID: 702); Glycine (PubChemCID:750); Lymphangiogenesis; Lymphedema; MAZ51, VEGF Receptor 3 Kinase inhibitor (PubChemCID:9839842); Methanol (PubChemCID:887); PD98059 (PubChemCID: 4713); SB203580 (PubChemCID: 176155); SP600125 (PubChemCID: 8515); Saponins of panaxnotoginseng; Sodium chloride (PubChemCID:5234); Vascular endothelial growth factors C; Wortmannin (PubChemCID: 312145).
Copyright © 2016 Elsevier Ireland Ltd. All rights reserved.
Conflict of interest statement
None of the authors have any competing interests in the manuscript. And this manuscript/data, or parts thereof, has not been submitted or published elsewhere for publication
Figures
Similar articles
-
Inhibiting adhesion events by Panax notoginseng saponins and Ginsenoside Rb1 protecting arteries via activation of Nrf2 and suppression of p38 - VCAM-1 signal pathway.J Ethnopharmacol. 2016 Nov 4;192:423-430. doi: 10.1016/j.jep.2016.09.022. Epub 2016 Sep 13. J Ethnopharmacol. 2016. PMID: 27620662
-
Src contributes to IL6-induced vascular endothelial growth factor-C expression in lymphatic endothelial cells.Angiogenesis. 2014 Apr;17(2):407-18. doi: 10.1007/s10456-013-9386-1. Epub 2013 Sep 19. Angiogenesis. 2014. PMID: 24048742
-
Interleukin-6 Induces Vascular Endothelial Growth Factor-C Expression via Src-FAK-STAT3 Signaling in Lymphatic Endothelial Cells.PLoS One. 2016 Jul 6;11(7):e0158839. doi: 10.1371/journal.pone.0158839. eCollection 2016. PLoS One. 2016. PMID: 27383632 Free PMC article.
-
Two Birds, One Stone: Double Hits on Tumor Growth and Lymphangiogenesis by Targeting Vascular Endothelial Growth Factor Receptor 3.Cells. 2019 Mar 21;8(3):270. doi: 10.3390/cells8030270. Cells. 2019. PMID: 30901976 Free PMC article. Review.
-
Dysregulation of Lymphatic Endothelial VEGFR3 Signaling in Disease.Cells. 2023 Dec 28;13(1):68. doi: 10.3390/cells13010068. Cells. 2023. PMID: 38201272 Free PMC article. Review.
Cited by
-
Lymphangiogenic responses of lymphatic endothelial cells to steady direct-current electric fields.Cell Adh Migr. 2023 Dec;17(1):1-14. doi: 10.1080/19336918.2023.2271260. Epub 2023 Oct 27. Cell Adh Migr. 2023. PMID: 37889090 Free PMC article.
-
Heteronemin Suppresses Lymphangiogenesis through ARF-1 and MMP-9/VE-Cadherin/Vimentin.Biomedicines. 2021 Aug 29;9(9):1109. doi: 10.3390/biomedicines9091109. Biomedicines. 2021. PMID: 34572295 Free PMC article.
-
Elevated ERβ expression driven by low ASB8-mediated ubiquitination in lung adenocarcinoma promotes lymph node metastasis via tumor-associated neutrophils.Cell Death Dis. 2025 Jul 30;16(1):576. doi: 10.1038/s41419-025-07870-z. Cell Death Dis. 2025. PMID: 40739091 Free PMC article.
-
Acquired lymphedema: Molecular contributors and future directions for developing intervention strategies.Front Pharmacol. 2022 Oct 25;13:873650. doi: 10.3389/fphar.2022.873650. eCollection 2022. Front Pharmacol. 2022. PMID: 36386144 Free PMC article. Review.
-
Esophageal carcinoma cell-excreted exosomal uc.189 promotes lymphatic metastasis.Aging (Albany NY). 2021 May 23;13(10):13846-13858. doi: 10.18632/aging.202979. Epub 2021 May 23. Aging (Albany NY). 2021. PMID: 34024769 Free PMC article.
References
-
- Beesley V, Janda M, Eakin E, Obermair A, Battistutta D. Lymphedema after gynecological cancer treatment: prevalence, correlates, and supportive care needs. Cancer. 2007;109:2607–2614. - PubMed
-
- Chen QS. Pharmacological studies on notoginseng saponins isolated from the fibrous root of Panax notoginseng. Zhong Yao Tong Bao. 1987;12:45–47. - PubMed
-
- Chen S, Liu J, Liu X, Fu Y, Zhang M, Lin Q, Zhu J, Mai L, Shan Z, Yu X, Yang M, Lin S. Panax notoginseng saponins inhibit ischemia-induced apoptosis by activating PI3K/Akt pathway in cardiomyocytes. Journal of ethnopharmacology. 2011;137:263–270. - PubMed
-
- Cheung L, Han J, Beilhack A, Joshi S, Wilburn P, Dua A, An A, Rockson SG. An experimental model for the study of lymphedema and its response to therapeutic lymphangiogenesis. BioDrugs: clinical immunotherapeutics, biopharmaceuticals and gene therapy. 2006;20:363–370. - PubMed
-
- Cheung LW, Leung KW, Wong CK, Wong RN, Wong AS. Ginsenoside-Rg1 induces angiogenesis via non-genomic crosstalk of glucocorticoid receptor and fibroblast growth factor receptor-1. Cardiovascular research. 2011;89:419–425. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

