Efficacy and safety of bevacizumab plus chemotherapy compared to chemotherapy alone in previously untreated advanced or metastatic colorectal cancer: a systematic review and meta-analysis
- PMID: 27558497
- PMCID: PMC4997727
- DOI: 10.1186/s12885-016-2734-y
Efficacy and safety of bevacizumab plus chemotherapy compared to chemotherapy alone in previously untreated advanced or metastatic colorectal cancer: a systematic review and meta-analysis
Abstract
Background: Colorectal cancer (CRC) is the fourth most frequently diagnosed cancer and the second leading cause of neoplasm-related death in the United States. Several studies analyzed the efficacy of bevacizumab combined with different chemotherapy regimens consisting on drugs such as 5-FU, capecitabine, irinotecan and oxaliplatin. This systematic review aims to evaluate the effectiveness and safety of chemotherapy plus bevacizumab versus chemotherapy alone in patients with previously untreated advanced or metastatic colorectal cancer (mCRC).
Methods: Several databases were searched, including MEDLINE, EMBASE, LILACS, and CENTRAL. The primary endpoints were overall survival and progression-free survival. Data extracted from the studies were combined by using hazard ratio (HR) or risk ratio (RR) with their corresponding 95 % confidence intervals (95 % CI).
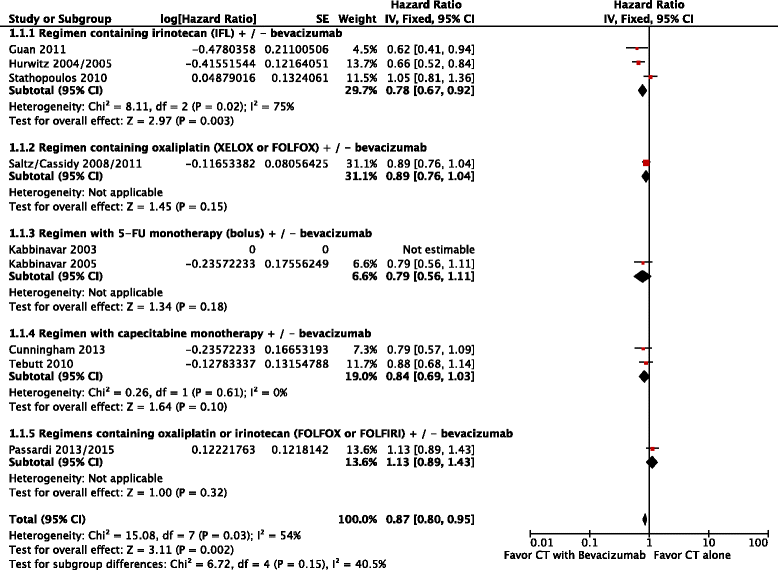
Results: The final analysis included 9 trials comprising 3,914 patients. Patients who received the combined treatment (chemotherapy + bevacizumab) had higher response rates (RR = 0.89; 95 % CI: 0.82 to 0.96; p = 0.003) with heterogeneity, higher progression-free survival (HR = 0.69; 95 % CI: 0.63 to 0.75; p < 0.00001) and also higher overall survival rates (HR = 0.87; 95 % CI: 0.80 to 0.95; p = 0.002) with moderate heterogeneity. Regarding adverse events and severe toxicities (grade ≥ 3), the group receiving the combined therapy had higher rates of hypertension (RR = 3.56 95 % CI: 2.58 to 4.92; p < 0.00001), proteinuria (RR = 1.89; 95 % CI: 1.26 to 2.84; p = 0.002), gastrointestinal perforation (RR = 3.63; 95 % CI: 1.31 to 10.09; p = 0.01), any thromboembolic events (RR = 1.44; 95 % CI: 1.20 to 1.73; p = 0.0001), and bleeding (RR = 1.81; 95 % CI: 1.22 to 2.67; p = 0.003).
Conclusion: The combination of chemotherapy with bevacizumab increased the response rate, progression-free survival and overall survival of patients with mCRC without prior chemotherapy. The results of progression-free survival (PFS) and overall survival (OS) were comparatively higher in those subgroups of patients receiving bolus 5-FU or capecitabine-based chemotherapy plus bevacizumab, when compared to patients treated with infusional %-FU plus bevacizumab (no difference in PFS and OS). Regarding the type of cytotoxic scheme, regimens containing irinotecan and fluoropyrimidine monotherapy showed superior efficacy results when combined to bevacizumab.
Keywords: Bevacizumab; Chemotherapy; Meta-analysis; Metastatic colorectal cancer; Systematic review.
Figures



References
-
- National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology: Colon Cancer (Version 2.2016). http://www.nccn.org/professionals/physician_gls/pdf/colon.pdf - PubMed
-
- Van Cutsem E, Oliveira J. Advanced colorectal cancer: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2009;20(Suppl 4):61–3. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

