Functional diversity of excitatory commissural interneurons in adult zebrafish
- PMID: 27559611
- PMCID: PMC5039025
- DOI: 10.7554/eLife.18579
Functional diversity of excitatory commissural interneurons in adult zebrafish
Abstract
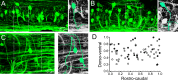
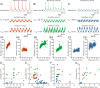
Flexibility in the bilateral coordination of muscle contraction underpins variable locomotor movements or gaits. While the locomotor rhythm is generated by ipsilateral excitatory interneurons, less is known about the commissural excitatory interneurons. Here we examined how the activity of the V0v interneurons - an important commissural neuronal class - varies with the locomotor speed in adult zebrafish. Although V0v interneurons are molecularly homogenous, their activity pattern during locomotion is not uniform. They consist of two distinct types dependent on whether they display rhythmicity or not during locomotion. The rhythmic V0v interneurons were further subdivided into three sub-classes engaged sequentially, first at slow then intermediate and finally fast locomotor speeds. Their order of recruitment is defined by scaling their synaptic current with their input resistance. Thus we uncover, in an adult vertebrate, a novel organizational principle for a key class of commissural interneurons and their recruitment pattern as a function of locomotor speed.
Keywords: interneurons; locomotion; neuroscience; spinal cord; zebrafish.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures



Similar articles
-
Organization of left-right coordination of neuronal activity in the mammalian spinal cord: Insights from computational modelling.J Physiol. 2015 Jun 1;593(11):2403-26. doi: 10.1113/JP270121. J Physiol. 2015. PMID: 25820677 Free PMC article.
-
Central control of interlimb coordination and speed-dependent gait expression in quadrupeds.J Physiol. 2016 Dec 1;594(23):6947-6967. doi: 10.1113/JP272787. Epub 2016 Nov 8. J Physiol. 2016. PMID: 27633893 Free PMC article.
-
Separate microcircuit modules of distinct v2a interneurons and motoneurons control the speed of locomotion.Neuron. 2014 Aug 20;83(4):934-43. doi: 10.1016/j.neuron.2014.07.018. Epub 2014 Aug 7. Neuron. 2014. PMID: 25123308
-
The intrinsic operation of the networks that make us locomote.Curr Opin Neurobiol. 2015 Apr;31:244-9. doi: 10.1016/j.conb.2015.01.003. Epub 2015 Jan 17. Curr Opin Neurobiol. 2015. PMID: 25599926 Review.
-
Diversity of molecularly defined spinal interneurons engaged in mammalian locomotor pattern generation.J Neurophysiol. 2017 Dec 1;118(6):2956-2974. doi: 10.1152/jn.00322.2017. Epub 2017 Aug 30. J Neurophysiol. 2017. PMID: 28855288 Free PMC article. Review.
Cited by
-
Delineating the Diversity of Spinal Interneurons in Locomotor Circuits.J Neurosci. 2017 Nov 8;37(45):10835-10841. doi: 10.1523/JNEUROSCI.1829-17.2017. J Neurosci. 2017. PMID: 29118212 Free PMC article. Review.
-
Caenorhabditis elegans excitatory ventral cord motor neurons derive rhythm for body undulation.Philos Trans R Soc Lond B Biol Sci. 2018 Sep 10;373(1758):20170370. doi: 10.1098/rstb.2017.0370. Philos Trans R Soc Lond B Biol Sci. 2018. PMID: 30201835 Free PMC article. Review.
-
Conserved neural circuit structure across Drosophila larval development revealed by comparative connectomics.Elife. 2017 Oct 23;6:e29089. doi: 10.7554/eLife.29089. Elife. 2017. PMID: 29058674 Free PMC article.
-
Principles Governing Locomotion in Vertebrates: Lessons From Zebrafish.Front Neural Circuits. 2018 Sep 13;12:73. doi: 10.3389/fncir.2018.00073. eCollection 2018. Front Neural Circuits. 2018. PMID: 30271327 Free PMC article. Review.
-
Behavioral Characterization of dmrt3a Mutant Zebrafish Reveals Crucial Aspects of Vertebrate Locomotion through Phenotypes Related to Acceleration.eNeuro. 2020 May 18;7(3):ENEURO.0047-20.2020. doi: 10.1523/ENEURO.0047-20.2020. Print 2020 May/Jun. eNeuro. 2020. PMID: 32357958 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

