Anti-Inflammatory Activity of Bee Venom in BV2 Microglial Cells: Mediation of MyD88-Dependent NF-κB Signaling Pathway
- PMID: 27563334
- PMCID: PMC4987476
- DOI: 10.1155/2016/3704764
Anti-Inflammatory Activity of Bee Venom in BV2 Microglial Cells: Mediation of MyD88-Dependent NF-κB Signaling Pathway
Abstract
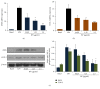
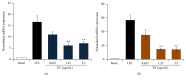
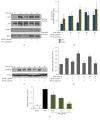
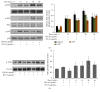
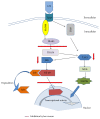
Bee venom has long been used as a traditional folk medicine in Korea. It has been reportedly used for the treatment of arthritis, cancer, and inflammation. Although its anti-inflammatory activity in lipopolysaccharide- (LPS-) stimulated inflammatory cells has been reported, the exact mechanism of its anti-inflammatory action has not been fully elucidated. Therefore, the aim of this study was to investigate the anti-inflammatory mechanism of bee venom in BV2 microglial cells. We first investigated whether NO production in LPS-activated BV2 cells was inhibited by bee venom, and further iNOS mRNA and protein expressions were determined. The mRNA and protein levels of proinflammatory cytokines were examined using semiquantitative RT-PCR and immunoblotting, respectively. Moreover, modulation of the transcription factor NF-κB by bee venom was also investigated using a luciferase assay. LPS-induced NO production in BV2 microglial cells was significantly inhibited in a concentration-dependent manner upon pretreatment with bee venom. Bee venom markedly reduced the mRNA expression of COX-2, TNF-α, IL-1β, and IL-6 and suppressed LPS-induced activation of MyD88 and IRAK1 and phosphorylation of TAK1. Moreover, NF-κB translocation by IKKα/β phosphorylation and subsequent IκB-α degradation were also attenuated. Thus, collectively, these results indicate that bee venom exerts its anti-inflammatory activity via the IRAK1/TAK1/NF-κB signaling pathway.
Figures


Similar articles
-
Quercetin disrupts tyrosine-phosphorylated phosphatidylinositol 3-kinase and myeloid differentiation factor-88 association, and inhibits MAPK/AP-1 and IKK/NF-κB-induced inflammatory mediators production in RAW 264.7 cells.Immunobiology. 2013 Dec;218(12):1452-67. doi: 10.1016/j.imbio.2013.04.019. Epub 2013 May 9. Immunobiology. 2013. PMID: 23735482
-
JNK pathway is involved in the inhibition of inflammatory target gene expression and NF-kappaB activation by melittin.J Inflamm (Lond). 2008 May 29;5:7. doi: 10.1186/1476-9255-5-7. J Inflamm (Lond). 2008. PMID: 18507870 Free PMC article.
-
Bee venom and melittin reduce proinflammatory mediators in lipopolysaccharide-stimulated BV2 microglia.Int Immunopharmacol. 2007 Aug;7(8):1092-101. doi: 10.1016/j.intimp.2007.04.005. Epub 2007 May 2. Int Immunopharmacol. 2007. PMID: 17570326
-
Therapeutic Potential of Bee and Wasp Venom in Anti-Arthritic Treatment: A Review.Toxins (Basel). 2024 Oct 22;16(11):452. doi: 10.3390/toxins16110452. Toxins (Basel). 2024. PMID: 39591207 Free PMC article. Review.
-
Honey Bee Products: Preclinical and Clinical Studies of Their Anti-inflammatory and Immunomodulatory Properties.Front Nutr. 2022 Jan 3;8:761267. doi: 10.3389/fnut.2021.761267. eCollection 2021. Front Nutr. 2022. PMID: 35047540 Free PMC article. Review.
Cited by
-
The Ameliorative Effects of the Ethyl Acetate Extract of Salicornia europaea L. and Its Bioactive Candidate, Irilin B, on LPS-Induced Microglial Inflammation and MPTP-Intoxicated PD-Like Mouse Model.Oxid Med Cell Longev. 2019 Jul 9;2019:6764756. doi: 10.1155/2019/6764756. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31379989 Free PMC article.
-
Anti-Neuroinflammatory Effect of the Ethanolic Extract of Black Ginseng through TLR4-MyD88-Regulated Inhibition of NF-κB and MAPK Signaling Pathways in LPS-Induced BV2 Microglial Cells.Int J Mol Sci. 2023 Oct 18;24(20):15320. doi: 10.3390/ijms242015320. Int J Mol Sci. 2023. PMID: 37894998 Free PMC article.
-
Bee Venom: From Venom to Drug.Molecules. 2021 Aug 15;26(16):4941. doi: 10.3390/molecules26164941. Molecules. 2021. PMID: 34443529 Free PMC article. Review.
-
Pharmacokinetic improvement provided by microneedle patch in delivering bee venom, a case study in combating scopolamine-induced neurodegeneration in mouse model.Drug Deliv. 2022 Dec;29(1):2855-2867. doi: 10.1080/10717544.2022.2116129. Drug Deliv. 2022. PMID: 36065088 Free PMC article.
-
Antiinflammatory peptides: current knowledge and promising prospects.Inflamm Res. 2019 Feb;68(2):125-145. doi: 10.1007/s00011-018-1208-x. Epub 2018 Dec 17. Inflamm Res. 2019. PMID: 30560372 Review.
References
-
- Guillott L., Medjane S., Le-Barillec K., et al. Response of human pulmonary epithelial cells to lipopolysaccharide involves toll-like receptor 4 (TLR4)-dependent signaling pathways: evidence for an intracellular compartmentalization of TLR4. The Journal of Biological Chemistry. 2004;279(4):2712–2718. doi: 10.1074/jbc.m305790200. - DOI - PubMed
-
- Gorina R., Font-Nieves M., Márquez-Kisinousky L., Santalucia T., Planas A. M. Astrocyte TLR4 activation induces a proinflammatory environment through the interplay between MyD88-dependent NFκB signaling, MAPK, and Jak1/Stat1 pathways. GLIA. 2011;59(2):242–255. doi: 10.1002/glia.21094. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

