TGFβ induces BIGH3 expression and human retinal pericyte apoptosis: a novel pathway of diabetic retinopathy
- PMID: 27564721
- PMCID: PMC5177749
- DOI: 10.1038/eye.2016.179
TGFβ induces BIGH3 expression and human retinal pericyte apoptosis: a novel pathway of diabetic retinopathy
Abstract
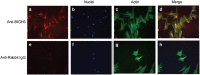
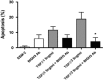
PurposeOne of the earliest hallmarks of diabetic retinopathy is the loss of retinal pericytes. However, the mechanisms that promote pericyte dropout are unknown. In the present study, we propose a novel pathway in which pericyte apoptosis is mediated by macrophages, TGFβ and pro-apoptotic BIGH3 (TGFβ-induced Gene Human Clone 3) protein.Patients and methodsTo elucidate this pathway, we assayed human retinal pericyte (HRP) apoptosis by TUNEL assay, BIGH3 mRNA expression by qPCR, and BIGH3 protein expression by western blot analysis. HRP were treated with BIGH3 protein, TGFβ1 and TGFβ2 and inhibition assays were carried out by blocking with antibodies against BIGH3. The distribution of BIGH3 and CD68+ macrophages were compared in a post-mortem donor eye with 7-year history of Type II diabetes and histopathogically confirmed non-proliferative diabetic retinopathy (NPDR).ResultsTGFβ induced a significant increase in BIGH3 mRNA and protein expression, and HRP apoptosis. BIGH3 treatment showed HRP undergo apoptosis in a dose-dependent manner. At 5 μg/ml, BIGH3 induced 3.5-times more apoptosis in HRP than in retinal endothelial cells. TGFβ induced apoptosis was inhibited by blocking with antibodies against BIGH3. In an example of NPDR, BIGH3 accumulated within the walls of the inner retina arterioles. Macrophage infiltrates were frequently associated with these vessels and the inner nuclear layer.ConclusionTogether with our previously published results on macrophage-induced retinal endothelial cell apoptosis, the present study supports a novel inflammatory pathway mediated by macrophages and the BIGH3 protein leading to HRP apoptosis. As shown in human post-mortem globes, these observations are clinically relevant, suggesting a new mechanism underlying pericyte dropout during NPDR.
Figures



Similar articles
-
BIGH3 protein and macrophages in retinal endothelial cell apoptosis.Apoptosis. 2015 Jan;20(1):29-37. doi: 10.1007/s10495-014-1052-6. Apoptosis. 2015. PMID: 25378215 Free PMC article.
-
Bax is increased in the retina of diabetic subjects and is associated with pericyte apoptosis in vivo and in vitro.Am J Pathol. 2000 Mar;156(3):1025-32. doi: 10.1016/S0002-9440(10)64970-X. Am J Pathol. 2000. PMID: 10702418 Free PMC article.
-
Imbalance between pro-apoptotic and pro-survival factors in human retinal pericytes in diabetic-like conditions.Acta Ophthalmol. 2018 Feb;96(1):e19-e26. doi: 10.1111/aos.13377. Epub 2017 Jan 27. Acta Ophthalmol. 2018. PMID: 28127871
-
Targeting pericyte retention in Diabetic Retinopathy: a review.Ann Med. 2024 Dec;56(1):2398200. doi: 10.1080/07853890.2024.2398200. Epub 2024 Sep 13. Ann Med. 2024. PMID: 39268600 Free PMC article. Review.
-
Importance of pericytes and mechanisms of pericyte loss during diabetes retinopathy.Diabetes Obes Metab. 2008 Jan;10(1):53-63. doi: 10.1111/j.1463-1326.2007.00795.x. Epub 2007 Oct 15. Diabetes Obes Metab. 2008. PMID: 17941874 Review.
Cited by
-
Genome-wide pathogenesis interpretation using a heat diffusion-based systems genetics method and implications for gene function annotation.Mol Genet Genomic Med. 2020 Oct;8(10):e1456. doi: 10.1002/mgg3.1456. Epub 2020 Sep 1. Mol Genet Genomic Med. 2020. PMID: 32869547 Free PMC article.
-
TGF-β Signaling Pathways in the Development of Diabetic Retinopathy.Int J Mol Sci. 2024 Mar 6;25(5):3052. doi: 10.3390/ijms25053052. Int J Mol Sci. 2024. PMID: 38474297 Free PMC article. Review.
-
Transcriptomic analysis of cells in response to EV71 infection and 2Apro as a trigger for apoptosis via TXNIP gene.Genes Genomics. 2019 Mar;41(3):343-357. doi: 10.1007/s13258-018-0760-7. Epub 2018 Nov 29. Genes Genomics. 2019. PMID: 30499052
-
Pyroptosis in the Retinal Neurovascular Unit: New Insights Into Diabetic Retinopathy.Front Immunol. 2021 Oct 19;12:763092. doi: 10.3389/fimmu.2021.763092. eCollection 2021. Front Immunol. 2021. PMID: 34737754 Free PMC article. Review.
-
Evolving cancer-niche interactions and therapeutic targets during bone metastasis.Nat Rev Cancer. 2022 Feb;22(2):85-101. doi: 10.1038/s41568-021-00406-5. Epub 2021 Oct 5. Nat Rev Cancer. 2022. PMID: 34611349 Free PMC article. Review.
References
-
- Geiss LS, Wang J, Cheng YJ, Thompson TJ, Barker L, Li Y et al. Prevalence and incidence trends for diagnosed diabetes among adults aged 20 to 79 years, United States, 1980-2012. JAMA 2014; 312(12): 1218–1226. - PubMed
-
- Kempen JH, O'Colmain BJ, Leske MC, Haffner SM, Klein R, Moss SE et al. The prevalence of diabetic retinopathy among adults in the United States. Arch Ophthalmol 2004; 122(4): 552–563. - PubMed
-
- Gardner TW, Antonetti DA, Barber AJ, LaNoue KF, Levison SW. Diabetic retinopathy: more than meets the eye. Surv Ophthalmol 2002; 47(Suppl 2): S253–S262. - PubMed
-
- Hall AP. Review of the pericyte during angiogenesis and its role in cancer and diabetic retinopathy. Toxicol Pathol 2006; 34(6): 763–775. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

