Etifoxine improves sensorimotor deficits and reduces glial activation, neuronal degeneration, and neuroinflammation in a rat model of traumatic brain injury
- PMID: 27565146
- PMCID: PMC5002207
- DOI: 10.1186/s12974-016-0687-3
Etifoxine improves sensorimotor deficits and reduces glial activation, neuronal degeneration, and neuroinflammation in a rat model of traumatic brain injury
Abstract
Background: Traumatic brain injury (TBI) results in important neurological impairments which occur through a cascade of deleterious physiological events over time. There are currently no effective treatments to prevent these consequences. TBI is followed not only by an inflammatory response but also by a profound reorganization of the GABAergic system and a dysregulation of translocator protein 18 kDa (TSPO). Etifoxine is an anxiolytic compound that belongs to the benzoxazine family. It potentiates GABAergic neurotransmission, either through a positive allosteric effect or indirectly, involving the activation of TSPO that leads to an increase in neurosteroids synthesis. In several models of peripheral nerve injury, etifoxine has been demonstrated to display potent regenerative and anti-inflammatory properties and to promote functional recovery. Prior study also showed etifoxine efficacy in reducing brain edema in rats. In light of these positive results, we used a rat model of TBI to explore etifoxine treatment effects in a central nervous system injury, from functional outcomes to the underlying mechanisms.
Methods: Male Sprague-Dawley rats received contusion (n = 18) or sham (n = 19) injuries centered laterally to bregma over the left sensorimotor cortex. They were treated with etifoxine (50 mg/kg, i.p.) or its vehicle 30 min following injury and every day during 7 days. Rats underwent behavioral testing to assess sensorimotor function. In another experiment, injured rats (n = 10) or sham rats (n = 10) received etifoxine (EFX) (50 mg/kg, i.p.) or its vehicle 30 min post-surgery. Brains were then dissected for analysis of neuroinflammation markers, glial activation, and neuronal degeneration.
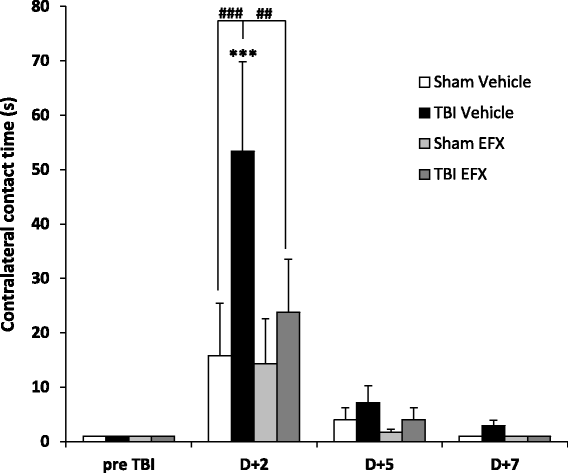
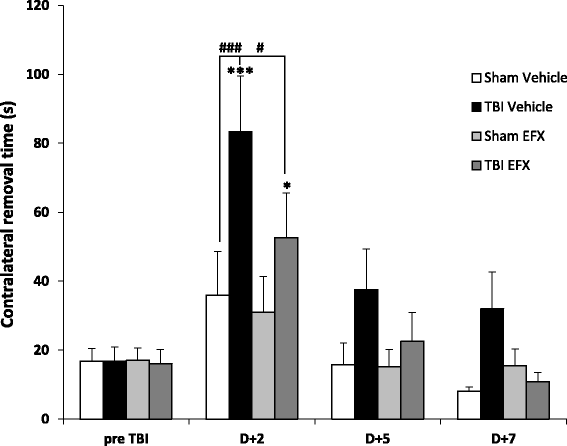
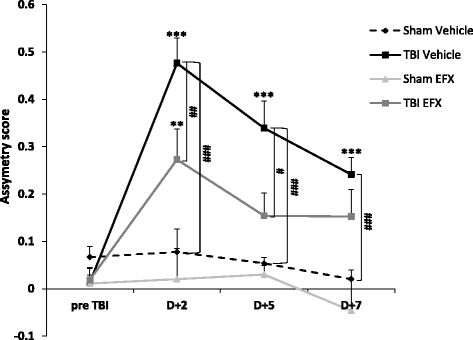
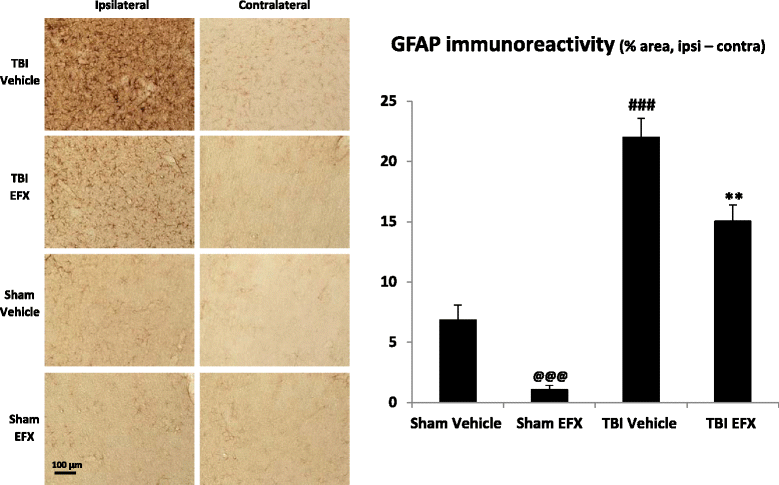
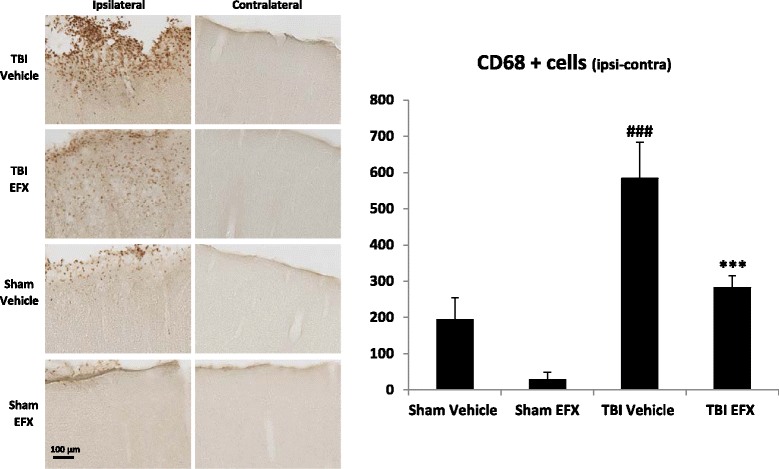
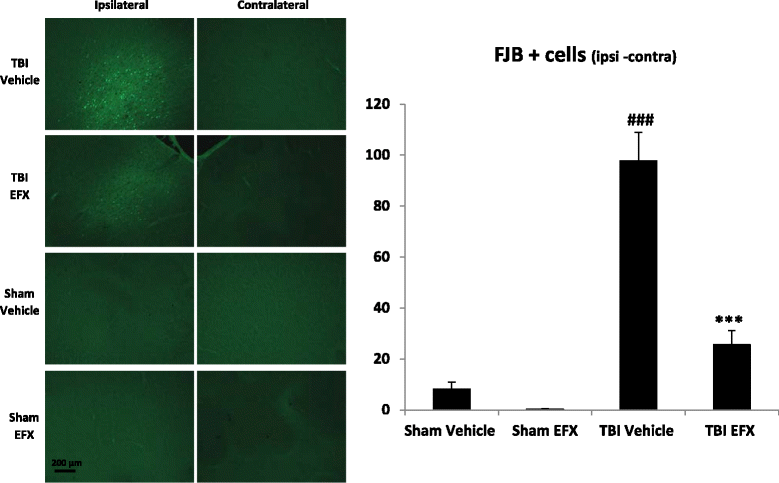
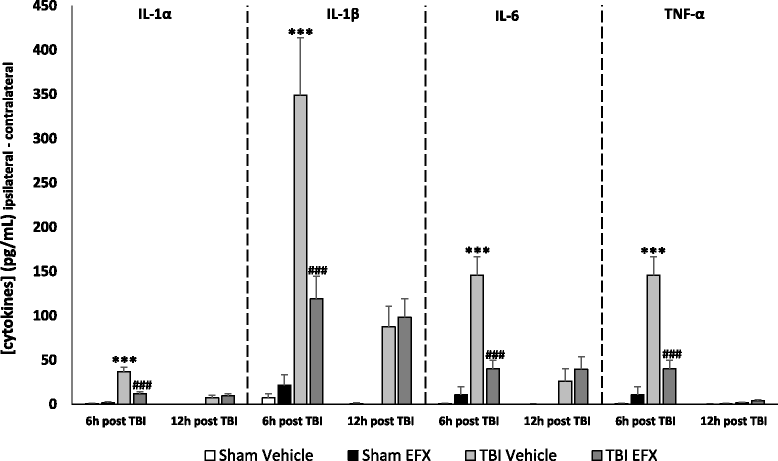
Results: Brain-injured rats exhibited significant sensorimotor function deficits compared to sham-injured rats in the bilateral tactile adhesive removal test, the beam walking test, and the limb-use asymmetry test. After 2 days of etifoxine treatment, behavioral impairments were significantly reduced. Etifoxine treatment reduced pro-inflammatory cytokines levels without affecting anti-inflammatory cytokines levels in injured rats, reduced macrophages and glial activation, and reduced neuronal degeneration.
Conclusions: Our results showed that post-injury treatment with etifoxine improved functional recovery and reduced neuroinflammation in a rat model of TBI. These findings suggest that etifoxine may have a therapeutic potential in the treatment of TBI.
Keywords: Astrogliosis; Cytokines; Etifoxine; Functional recovery; Microglia; Neuroinflammation; Neuronal degeneration; Neurosteroids; TSPO; Traumatic brain injury.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

