The novel mitochondria-targeted hydrogen sulfide (H2S) donors AP123 and AP39 protect against hyperglycemic injury in microvascular endothelial cells in vitro
- PMID: 27565382
- PMCID: PMC5113977
- DOI: 10.1016/j.phrs.2016.08.019
The novel mitochondria-targeted hydrogen sulfide (H2S) donors AP123 and AP39 protect against hyperglycemic injury in microvascular endothelial cells in vitro
Abstract
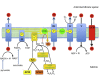
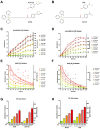
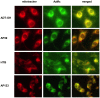
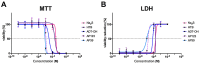
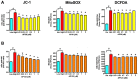
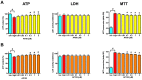
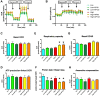
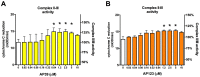
The development of diabetic vascular complications is initiated, at least in part, by mitochondrial reactive oxygen species (ROS) production in endothelial cells. Hyperglycemia induces superoxide production in the mitochondria and initiates changes in the mitochondrial membrane potential that leads to mitochondrial dysfunction. Hydrogen sulfide (H2S) supplementation has been shown to reduce the mitochondrial oxidant production and shows efficacy against diabetic vascular damage in vivo. However, the half-life of H2S is very short and it is not specific for the mitochondria. We have therefore evaluated two novel mitochondria-targeted anethole dithiolethione and hydroxythiobenzamide H2S donors (AP39 and AP123 respectively) at preventing hyperglycemia-induced oxidative stress and metabolic changes in microvascular endothelial cells in vitro. Hyperglycemia (HG) induced significant increase in the activity of the citric acid cycle and led to elevated mitochondrial membrane potential. Mitochondrial oxidant production was increased and the mitochondrial electron transport decreased in hyperglycemic cells. AP39 and AP123 (30-300nM) decreased HG-induced hyperpolarisation of the mitochondrial membrane and inhibited the mitochondrial oxidant production. Both H2S donors (30-300nM) increased the electron transport at respiratory complex III and improved the cellular metabolism. Targeting H2S to mitochondria retained the cytoprotective effect of H2S against glucose-induced damage in endothelial cells suggesting that the molecular target of H2S action is within the mitochondria. Mitochondrial targeting of H2S also induced >1000-fold increase in the potency of H2S against hyperglycemia-induced injury. The high potency and long-lasting effect elicited by these H2S donors strongly suggests that these compounds could be useful against diabetic vascular complications.
Keywords: Bioenergetics; Complex II; Electron transport; Endothelial cells; Hydrogen sulfide; Hyperglycemia; Oxidative stress; SQR; Superoxide.
Copyright © 2016 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures
Similar articles
-
AP39, a novel mitochondria-targeted hydrogen sulfide donor, stimulates cellular bioenergetics, exerts cytoprotective effects and protects against the loss of mitochondrial DNA integrity in oxidatively stressed endothelial cells in vitro.Nitric Oxide. 2014 Sep 15;41:120-30. doi: 10.1016/j.niox.2014.04.008. Epub 2014 Apr 19. Nitric Oxide. 2014. PMID: 24755204 Free PMC article.
-
Regulation of Vascular Tone, Angiogenesis and Cellular Bioenergetics by the 3-Mercaptopyruvate Sulfurtransferase/H2S Pathway: Functional Impairment by Hyperglycemia and Restoration by DL-α-Lipoic Acid.Mol Med. 2015 Feb 18;21(1):1-14. doi: 10.2119/molmed.2015.00035. Mol Med. 2015. PMID: 25715337 Free PMC article.
-
Hydrogen sulfide replacement therapy protects the vascular endothelium in hyperglycemia by preserving mitochondrial function.Proc Natl Acad Sci U S A. 2011 Aug 16;108(33):13829-34. doi: 10.1073/pnas.1105121108. Epub 2011 Aug 1. Proc Natl Acad Sci U S A. 2011. PMID: 21808008 Free PMC article.
-
Targeting Mitochondrial Dysfunction to Prevent Endothelial Dysfunction and Atherosclerosis in Diabetes: Focus on the Novel Uncoupler BAM15.Int J Mol Sci. 2025 May 11;26(10):4603. doi: 10.3390/ijms26104603. Int J Mol Sci. 2025. PMID: 40429748 Free PMC article. Review.
-
Hyperglycemia-associated alterations in cellular signaling and dysregulated mitochondrial bioenergetics in human metabolic disorders.Eur J Nutr. 2016 Dec;55(8):2339-2345. doi: 10.1007/s00394-016-1212-2. Epub 2016 Apr 15. Eur J Nutr. 2016. PMID: 27084094 Free PMC article. Review.
Cited by
-
Biological Functions of Hydrogen Sulfide in Plants.Int J Mol Sci. 2022 Dec 1;23(23):15107. doi: 10.3390/ijms232315107. Int J Mol Sci. 2022. PMID: 36499443 Free PMC article. Review.
-
Hydrogen sulfide signaling in mitochondria and disease.FASEB J. 2019 Dec;33(12):13098-13125. doi: 10.1096/fj.201901304R. Epub 2019 Oct 24. FASEB J. 2019. PMID: 31648556 Free PMC article. Review.
-
Static Cold Storage with Mitochondria-Targeted Hydrogen Sulfide Donor Improves Renal Graft Function in an Ex Vivo Porcine Model of Controlled Donation-after-Cardiac-Death Kidney Transplantation.Int J Mol Sci. 2023 Sep 13;24(18):14017. doi: 10.3390/ijms241814017. Int J Mol Sci. 2023. PMID: 37762319 Free PMC article.
-
Duchenne's muscular dystrophy involves a defective transsulfuration pathway activity.Redox Biol. 2021 Sep;45:102040. doi: 10.1016/j.redox.2021.102040. Epub 2021 Jun 19. Redox Biol. 2021. PMID: 34174560 Free PMC article.
-
H2S Donors with Cytoprotective Effects in Models of MI/R Injury and Chemotherapy-Induced Cardiotoxicity.Antioxidants (Basel). 2023 Mar 5;12(3):650. doi: 10.3390/antiox12030650. Antioxidants (Basel). 2023. PMID: 36978898 Free PMC article. Review.
References
-
- Hex N., Bartlett C., Wright D., Taylor M., Varley D. Estimating the current and future costs of Type 1 and Type 2 diabetes in the UK, including direct health costs and indirect societal and productivity costs. Diabet. Med.: J. Br. Diabet. Assoc. 2012;29(7):855–862. (PubMed PMID: 22537247) - PubMed
-
- World Health Organization . Definition and diagnosis of diabetes mellitus and intermediate hyperglycaemia: report of a WHO/IDF consultation. World Health Organization; Geneva: 2006. International Diabetes Federation.
-
- Giorgino F., Leonardini A., Laviola L. Cardiovascular disease and glycemic control in type 2 diabetes: now that the dust is settling from large clinical trials. Ann. N. Y. Acad. Sci. 2013;1281:36–50. doi: 10.1111/nyas.12044. (PubMed PMID: 23387439 PubMed Central PMCID: PMC3715107) - DOI - PMC - PubMed
-
- Avogaro A. Is glycemic control relevant to cardiovascular clinical outcomes? Heart Metab. 2015;68:20–26.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

