JNK Phosphorylates SIRT6 to Stimulate DNA Double-Strand Break Repair in Response to Oxidative Stress by Recruiting PARP1 to DNA Breaks
- PMID: 27568560
- PMCID: PMC5089070
- DOI: 10.1016/j.celrep.2016.08.006
JNK Phosphorylates SIRT6 to Stimulate DNA Double-Strand Break Repair in Response to Oxidative Stress by Recruiting PARP1 to DNA Breaks
Abstract
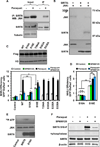
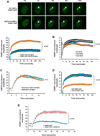
The accumulation of damage caused by oxidative stress has been linked to aging and to the etiology of numerous age-related diseases. The longevity gene, sirtuin 6 (SIRT6), promotes genome stability by facilitating DNA repair, especially under oxidative stress conditions. Here we uncover the mechanism by which SIRT6 is activated by oxidative stress to promote DNA double-strand break (DSB) repair. We show that the stress-activated protein kinase, c-Jun N-terminal kinase (JNK), phosphorylates SIRT6 on serine 10 in response to oxidative stress. This post-translational modification facilitates the mobilization of SIRT6 to DNA damage sites and is required for efficient recruitment of poly (ADP-ribose) polymerase 1 (PARP1) to DNA break sites and for efficient repair of DSBs. Our results demonstrate a post-translational mechanism regulating SIRT6, and they provide the link between oxidative stress signaling and DNA repair pathways that may be critical for hormetic response and longevity assurance.
Copyright © 2016 The Author(s). Published by Elsevier Inc. All rights reserved.
Figures
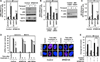

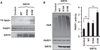
Similar articles
-
SIRT6 promotes DNA repair under stress by activating PARP1.Science. 2011 Jun 17;332(6036):1443-6. doi: 10.1126/science.1202723. Science. 2011. PMID: 21680843 Free PMC article.
-
Repairing split ends: SIRT6, mono-ADP ribosylation and DNA repair.Aging (Albany NY). 2011 Sep;3(9):829-35. doi: 10.18632/aging.100389. Aging (Albany NY). 2011. PMID: 21946623 Free PMC article. Review.
-
SIRT6 is a DNA double-strand break sensor.Elife. 2020 Jan 29;9:e51636. doi: 10.7554/eLife.51636. Elife. 2020. PMID: 31995034 Free PMC article.
-
Interaction of hepatitis B virus X protein with PARP1 results in inhibition of DNA repair in hepatocellular carcinoma.Oncogene. 2016 Oct 13;35(41):5435-5445. doi: 10.1038/onc.2016.82. Epub 2016 Apr 4. Oncogene. 2016. PMID: 27041572
-
The histone deacetylase SIRT6: at the crossroads between epigenetics, metabolism and disease.Curr Top Med Chem. 2013;13(23):2991-3000. doi: 10.2174/15680266113136660213. Curr Top Med Chem. 2013. PMID: 24171769 Review.
Cited by
-
SIRT6 in Senescence and Aging-Related Cardiovascular Diseases.Front Cell Dev Biol. 2021 Mar 29;9:641315. doi: 10.3389/fcell.2021.641315. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 33855020 Free PMC article. Review.
-
Natural Bioactive Compounds Targeting Histone Deacetylases in Human Cancers: Recent Updates.Molecules. 2022 Apr 15;27(8):2568. doi: 10.3390/molecules27082568. Molecules. 2022. PMID: 35458763 Free PMC article. Review.
-
SIRT6, a Mammalian Deacylase with Multitasking Abilities.Physiol Rev. 2020 Jan 1;100(1):145-169. doi: 10.1152/physrev.00030.2018. Epub 2019 Aug 22. Physiol Rev. 2020. PMID: 31437090 Free PMC article. Review.
-
Excess growth hormone suppresses DNA damage repair in epithelial cells.JCI Insight. 2019 Feb 7;4(3):e125762. doi: 10.1172/jci.insight.125762. JCI Insight. 2019. PMID: 30728323 Free PMC article.
-
Emerging Therapeutic Potential of SIRT6 Modulators.J Med Chem. 2021 Jul 22;64(14):9732-9758. doi: 10.1021/acs.jmedchem.1c00601. Epub 2021 Jul 2. J Med Chem. 2021. PMID: 34213345 Free PMC article. Review.
References
-
- Bergamini CM, Gambetti S, Dondi A, Cervellati C. Oxygen, reactive oxygen species and tissue damage. Curr. Pharm. Des. 2004;10:1611–1626. - PubMed
-
- Davis RJ. Signal transduction by the JNK group of MAP kinases. Cell. 2000;103:239–252. - PubMed
-
- Ford J, Ahmed S, Allison S, Jiang M, Milner J. JNK2-dependent regulation of SIRT1 protein stability. Cell Cycle. 2008;7:3091–3097. - PubMed
-
- Gertler AA, Cohen HY. SIRT6, a protein with many faces. Biogerontology. 2013;14:629–639. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

