Immune-Stimulatory Effects of Rapamycin Are Mediated by Stimulation of Antitumor γδ T Cells
- PMID: 27569211
- PMCID: PMC5065775
- DOI: 10.1158/0008-5472.CAN-16-0091
Immune-Stimulatory Effects of Rapamycin Are Mediated by Stimulation of Antitumor γδ T Cells
Abstract
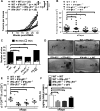
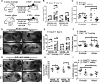
The FDA-approved mTOR inhibitor rapamycin mediates important immune effects, but its contributions to the anticancer effects of the drug are unclear. Here we report evidence that rapamycin-mediated cancer protection relies upon stimulation of γδ T cells. In a well-established mouse model of carcinogen and inflammation-driven skin carcinogenesis, IFNγ recruited γδ TCRmid T cells to the epidermis where rapamycin boosted their perforin-dependent antitumor properties. These antitumor cells were mostly Vγ5-Vγ4-Vγ1- in phenotype. IFNγ signals were required in both hematopoietic and nonhematopoietic cells for rapamycin to optimally promote epidermal infiltration of γδ TCRmid T cells, as mediated by CXCR3-CXCL10 interactions, along with the antitumor effects of these cells. In mouse xenograft models of human squamous cell carcinoma, rapamycin improved human γδ T-cell-mediated cancer cell killing. Our results identify immune mechanisms for the cancer prevention and treatment properties of rapamycin, challenging the paradigm that mTOR inhibition acts primarily by direct action on tumor cells. Cancer Res; 76(20); 5970-82. ©2016 AACR.
©2016 American Association for Cancer Research.
Figures





References
-
- Chiarini F, Evangelisti C, McCubrey JA, Martelli AM. Current treatment strategies for inhibiting mTOR in cancer. Trends Pharmacol Sci. 2015;36(2):124–35. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

