γδ T Cells Support Pancreatic Oncogenesis by Restraining αβ T Cell Activation
- PMID: 27569912
- PMCID: PMC5017923
- DOI: 10.1016/j.cell.2016.07.046
γδ T Cells Support Pancreatic Oncogenesis by Restraining αβ T Cell Activation
Erratum in
-
γδ T Cells Support Pancreatic Oncogenesis by Restraining αβ T Cell Activation.Cell. 2020 Nov 12;183(4):1134-1136. doi: 10.1016/j.cell.2020.10.041. Cell. 2020. PMID: 33186522 No abstract available.
Abstract
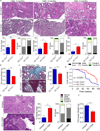
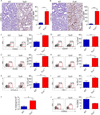
Inflammation is paramount in pancreatic oncogenesis. We identified a uniquely activated γδT cell population, which constituted ∼40% of tumor-infiltrating T cells in human pancreatic ductal adenocarcinoma (PDA). Recruitment and activation of γδT cells was contingent on diverse chemokine signals. Deletion, depletion, or blockade of γδT cell recruitment was protective against PDA and resulted in increased infiltration, activation, and Th1 polarization of αβT cells. Although αβT cells were dispensable to outcome in PDA, they became indispensable mediators of tumor protection upon γδT cell ablation. PDA-infiltrating γδT cells expressed high levels of exhaustion ligands and thereby negated adaptive anti-tumor immunity. Blockade of PD-L1 in γδT cells enhanced CD4(+) and CD8(+) T cell infiltration and immunogenicity and induced tumor protection suggesting that γδT cells are critical sources of immune-suppressive checkpoint ligands in PDA. We describe γδT cells as central regulators of effector T cell activation in cancer via novel cross-talk.
Keywords: Kras; cancer; checkpoint ligands.
Copyright © 2016 Elsevier Inc. All rights reserved.
Figures





Comment in
-
Pancreatic cancer: γδ T cells support PDAC.Nat Rev Gastroenterol Hepatol. 2016 Oct;13(10):560. doi: 10.1038/nrgastro.2016.156. Epub 2016 Sep 14. Nat Rev Gastroenterol Hepatol. 2016. PMID: 27625194 No abstract available.
References
-
- Andren-Sandberg A, Dervenis C, Lowenfels B. Etiologic links between chronic pancreatitis and pancreatic cancer. Scand J Gastroenterol. 1997;32:97–103. - PubMed
-
- Clark CE, Hingorani SR, Mick R, Combs C, Tuveson DA, Vonderheide RH. Dynamics of the immune reaction to pancreatic cancer from inception to invasion. Cancer research. 2007;67:9518–9527. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

