Perineuronal Nets Enhance the Excitability of Fast-Spiking Neurons
- PMID: 27570824
- PMCID: PMC4987413
- DOI: 10.1523/ENEURO.0112-16.2016
Perineuronal Nets Enhance the Excitability of Fast-Spiking Neurons
Abstract
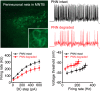
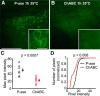
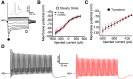
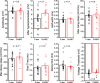
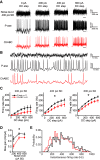
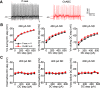
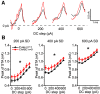
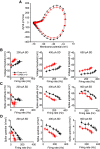
Perineuronal nets (PNNs) are specialized complexes of extracellular matrix molecules that surround the somata of fast-spiking neurons throughout the vertebrate brain. PNNs are particularly prevalent throughout the auditory brainstem, which transmits signals with high speed and precision. It is unknown whether PNNs contribute to the fast-spiking ability of the neurons they surround. Whole-cell recordings were made from medial nucleus of the trapezoid body (MNTB) principal neurons in acute brain slices from postnatal day 21 (P21) to P27 mice. PNNs were degraded by incubating slices in chondroitinase ABC (ChABC) and were compared to slices that were treated with a control enzyme (penicillinase). ChABC treatment did not affect the ability of MNTB neurons to fire at up to 1000 Hz when driven by current pulses. However, f-I (frequency-intensity) curves constructed by injecting Gaussian white noise currents superimposed on DC current steps showed that ChABC treatment reduced the gain of spike output. An increase in spike threshold may have contributed to this effect, which is consistent with the observation that spikes in ChABC-treated cells were delayed relative to control-treated cells. In addition, parvalbumin-expressing fast-spiking cortical neurons in >P70 slices that were treated with ChABC also had reduced excitability and gain. The development of PNNs around somata of fast-spiking neurons may be essential for fast and precise sensory transmission and synaptic inhibition in the brain.
Keywords: auditory brainstem; extracellular matrix; fast spiking; inhibition; perineuronal net; white noise current.
Figures


References
-
- Bertolotto A, Manzardo E, Guglielmone R (1996) Immunohistochemical mapping of perineuronal nets containing chondroitin unsulfated proteoglycan in the rat central nervous system. Cell Tissue Res 283:283-295. - PubMed
-
- Blosa M, Sonntag M, Brückner G, Jäger C, Seeger G, Matthews RT, Rübsamen R, Arendt T, Morawski M (2013) Unique features of extracellular matrix in the mouse medial nucleus of trapezoid body—implications for physiological functions. Neuroscience 228:215-234. 10.1016/j.neuroscience.2012.10.003 - DOI - PubMed
-
- Blosa M, Sonntag M, Jäger C, Weigel S, Seeger J, Frischknecht R, Seidenbecher CI, Matthews RT, Arendt T, Rübsamen R, Morawski M (2015) The extracellular matrix molecule brevican is an integral component of the machinery mediating fast synaptic transmission at the calyx of Held. J Physiol 593:4341-4360. 10.1113/JP270849 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
