GPR88 in A2AR Neurons Enhances Anxiety-Like Behaviors
- PMID: 27570825
- PMCID: PMC4987659
- DOI: 10.1523/ENEURO.0202-16.2016
GPR88 in A2AR Neurons Enhances Anxiety-Like Behaviors
Abstract
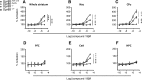
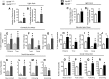
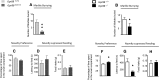
GPR88 is an orphan G-protein-coupled receptor highly expressed in striatal dopamine D1 (receptor) R- and D2R-expressing medium spiny neurons. This receptor is involved in activity and motor responses, and we previously showed that this receptor also regulates anxiety-like behaviors. To determine whether GPR88 in D2R-expressing neurons contributes to this emotional phenotype, we generated conditional Gpr88 knock-out mice using adenosine A2AR (A2AR)-Cre-driven recombination, and compared anxiety-related responses in both total and A2AR-Gpr88 KO mice. A2AR-Gpr88 KO mice showed a selective reduction of Gpr88 mRNA in D2R-expressing, but not D1R-expressing, neurons. These mutant mice showed increased locomotor activity and decreased anxiety-like behaviors in light/dark and elevated plus maze tests. These phenotypes were superimposable on those observed in total Gpr88 KO mice, demonstrating that the previously reported anxiogenic activity of GPR88 operates at the level of A2AR-expressing neurons. Further, A2AR-Gpr88 KO mice showed no change in novelty preference and novelty-suppressed feeding, while these responses were increased and decreased, respectively, in the total Gpr88 KO mice. Also, A2AR-Gpr88 KO mice showed intact fear conditioning, while the fear responses were decreased in total Gpr88 KO. We therefore also show for the first time that GPR88 activity regulates approach behaviors and conditional fear; however, these behaviors do not seem mediated by receptors in A2AR neurons. We conclude that Gpr88 expressed in A2AR neurons enhances ethological anxiety-like behaviors without affecting conflict anxiety and fear responses.
Keywords: D2R-medium spiny neurons; G-protein-coupled receptors; amygdale; anxiety-like behavior; striatum.
Figures

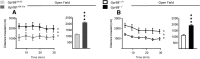
Similar articles
-
GPR88 in D1R-Type and D2R-Type Medium Spiny Neurons Differentially Regulates Affective and Motor Behavior.eNeuro. 2019 Aug 8;6(4):ENEURO.0035-19.2019. doi: 10.1523/ENEURO.0035-19.2019. Print 2019 Jul/Aug. eNeuro. 2019. PMID: 31346000 Free PMC article.
-
GPR88 in A2A receptor-expressing neurons modulates locomotor response to dopamine agonists but not sensorimotor gating.Eur J Neurosci. 2017 Aug;46(4):2026-2034. doi: 10.1111/ejn.13646. Epub 2017 Aug 2. Eur J Neurosci. 2017. PMID: 28700108 Free PMC article.
-
Mice Lacking GPR88 Show Motor Deficit, Improved Spatial Learning, and Low Anxiety Reversed by Delta Opioid Antagonist.Biol Psychiatry. 2016 Jun 1;79(11):917-27. doi: 10.1016/j.biopsych.2015.05.020. Epub 2015 Jun 6. Biol Psychiatry. 2016. PMID: 26188600 Free PMC article.
-
Allosteric Interactions between Adenosine A2A and Dopamine D2 Receptors in Heteromeric Complexes: Biochemical and Pharmacological Characteristics, and Opportunities for PET Imaging.Int J Mol Sci. 2021 Feb 9;22(4):1719. doi: 10.3390/ijms22041719. Int J Mol Sci. 2021. PMID: 33572077 Free PMC article. Review.
-
Allosteric mechanisms within the adenosine A2A-dopamine D2 receptor heterotetramer.Neuropharmacology. 2016 May;104:154-60. doi: 10.1016/j.neuropharm.2015.05.028. Epub 2015 Jun 4. Neuropharmacology. 2016. PMID: 26051403 Free PMC article. Review.
Cited by
-
Discovery of a Potent, Selective, and Brain-Penetrant Small Molecule that Activates the Orphan Receptor GPR88 and Reduces Alcohol Intake.J Med Chem. 2018 Aug 9;61(15):6748-6758. doi: 10.1021/acs.jmedchem.8b00566. Epub 2018 Jul 30. J Med Chem. 2018. PMID: 30011199 Free PMC article.
-
Orphan Receptor GPR88 as an Emerging Neurotherapeutic Target.ACS Chem Neurosci. 2019 Jan 16;10(1):190-200. doi: 10.1021/acschemneuro.8b00572. Epub 2018 Dec 20. ACS Chem Neurosci. 2019. PMID: 30540906 Free PMC article. Review.
-
Neuro-psychopharmacological perspective of Orphan receptors of Rhodopsin (class A) family of G protein-coupled receptors.Psychopharmacology (Berl). 2017 Apr;234(8):1181-1207. doi: 10.1007/s00213-017-4586-9. Epub 2017 Mar 13. Psychopharmacology (Berl). 2017. PMID: 28289782 Review.
-
Control of aversion by glycine-gated GluN1/GluN3A NMDA receptors in the adult medial habenula.Science. 2019 Oct 11;366(6462):250-254. doi: 10.1126/science.aax1522. Science. 2019. PMID: 31601771 Free PMC article.
-
Orphan peptide and G protein-coupled receptor signalling in alcohol use disorder.Br J Pharmacol. 2024 Mar;181(5):595-609. doi: 10.1111/bph.16301. Epub 2024 Jan 8. Br J Pharmacol. 2024. PMID: 38073127 Free PMC article. Review.
References
-
- Bailey KR, Crawley JN (2009) Anxiety- related behaviors in mice In: Methods of behavior analysis in neuroscience, Chap 5, Ed 2 (Buccafusco JJ, ed). Boca Raton, FL: CRC.
-
- Böhm C, Newrzella D, Herberger S, Schramm N, Eisenhardt G, Schenk V, Sonntag-Buck V, Sorgenfrei O (2006) Effects of antidepressant treatment on gene expression profile in mouse brain: cell type-specific transcription profiling using laser microdissection and microarray analysis. J Neurochem 97 [Suppl 1]:44–49. 10.1111/j.1471-4159.2006.03750.x - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
