Antibody-Mediated Internalization of Infectious HIV-1 Virions Differs among Antibody Isotypes and Subclasses
- PMID: 27579713
- PMCID: PMC5007037
- DOI: 10.1371/journal.ppat.1005817
Antibody-Mediated Internalization of Infectious HIV-1 Virions Differs among Antibody Isotypes and Subclasses
Abstract
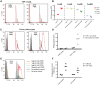
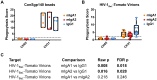
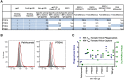
Emerging data support a role for antibody Fc-mediated antiviral activity in vaccine efficacy and in the control of HIV-1 replication by broadly neutralizing antibodies. Antibody-mediated virus internalization is an Fc-mediated function that may act at the portal of entry whereby effector cells may be triggered by pre-existing antibodies to prevent HIV-1 acquisition. Understanding the capacity of HIV-1 antibodies in mediating internalization of HIV-1 virions by primary monocytes is critical to understanding their full antiviral potency. Antibody isotypes/subclasses differ in functional profile, with consequences for their antiviral activity. For instance, in the RV144 vaccine trial that achieved partial efficacy, Env IgA correlated with increased risk of HIV-1 infection (i.e. decreased vaccine efficacy), whereas V1-V2 IgG3 correlated with decreased risk of HIV-1 infection (i.e. increased vaccine efficacy). Thus, understanding the different functional attributes of HIV-1 specific IgG1, IgG3 and IgA antibodies will help define the mechanisms of immune protection. Here, we utilized an in vitro flow cytometric method utilizing primary monocytes as phagocytes and infectious HIV-1 virions as targets to determine the capacity of Env IgA (IgA1, IgA2), IgG1 and IgG3 antibodies to mediate HIV-1 infectious virion internalization. Importantly, both broadly neutralizing antibodies (i.e. PG9, 2G12, CH31, VRC01 IgG) and non-broadly neutralizing antibodies (i.e. 7B2 mAb, mucosal HIV-1+ IgG) mediated internalization of HIV-1 virions. Furthermore, we found that Env IgG3 of multiple specificities (i.e. CD4bs, V1-V2 and gp41) mediated increased infectious virion internalization over Env IgG1 of the same specificity, while Env IgA mediated decreased infectious virion internalization compared to IgG1. These data demonstrate that antibody-mediated internalization of HIV-1 virions depends on antibody specificity and isotype. Evaluation of the phagocytic potency of vaccine-induced antibodies and therapeutic antibodies will enable a better understanding of their capacity to prevent and/or control HIV-1 infection in vivo.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

