Bone Morphogenetic Protein-2 Promotes Human Mesenchymal Stem Cell Survival and Resultant Bone Formation When Entrapped in Photocrosslinked Alginate Hydrogels
- PMID: 27581621
- PMCID: PMC5176258
- DOI: 10.1002/adhm.201600461
Bone Morphogenetic Protein-2 Promotes Human Mesenchymal Stem Cell Survival and Resultant Bone Formation When Entrapped in Photocrosslinked Alginate Hydrogels
Abstract
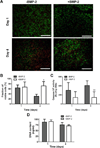
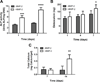
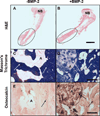
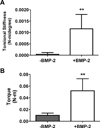
There is a substantial need to prolong cell persistence and enhance functionality in situ to enhance cell-based tissue repair. Bone morphogenetic protein-2 (BMP-2) is often used at high concentrations for osteogenic differentiation of mesenchymal stem cells (MSCs) but can induce apoptosis. Biomaterials facilitate the delivery of lower doses of BMP-2, reducing side effects and localizing materials at target sites. Photocrosslinked alginate hydrogels (PAHs) can deliver osteogenic materials to irregular-sized bone defects, providing improved control over material degradation compared to ionically cross-linked hydrogels. It is hypothesized that the delivery of MSCs and BMP-2 from a PAH increases cell persistence by reducing apoptosis, while promoting osteogenic differentiation and enhancing bone formation compared to MSCs in PAHs without BMP-2. BMP-2 significantly decreases apoptosis and enhances survival of photoencapsulated MSCs, while simultaneously promoting osteogenic differentiation in vitro. Bioluminescence imaging reveals increased MSC survival when implanted in BMP-2 PAHs. Bone defects treated with MSCs in BMP-2 PAHs demonstrate 100% union as early as 8 weeks and significantly higher bone volumes at 12 weeks, while defects with MSC-entrapped PAHs alone do not fully bridge. This study demonstrates that transplantation of MSCs with BMP-2 in PAHs achieves robust bone healing, providing a promising platform for bone repair.
Keywords: bone morphogenetic protein-2; mesenchymal stem cell; osteogenesis; photocrosslinking; survival.
© 2016 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
Figures

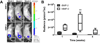

Similar articles
-
Effect of cell origin and timing of delivery for stem cell-based bone tissue engineering using biologically functionalized hydrogels.Tissue Eng Part A. 2015 Jan;21(1-2):156-65. doi: 10.1089/ten.TEA.2014.0057. Epub 2014 Aug 19. Tissue Eng Part A. 2015. PMID: 25010532 Free PMC article.
-
The role of BMP-7 in chondrogenic and osteogenic differentiation of human bone marrow multipotent mesenchymal stromal cells in vitro.J Cell Biochem. 2010 Feb 1;109(2):406-16. doi: 10.1002/jcb.22412. J Cell Biochem. 2010. PMID: 19950204
-
Increased Survival and Function of Mesenchymal Stem Cell Spheroids Entrapped in Instructive Alginate Hydrogels.Stem Cells Transl Med. 2016 Jun;5(6):773-81. doi: 10.5966/sctm.2015-0211. Epub 2016 Apr 7. Stem Cells Transl Med. 2016. PMID: 27057004 Free PMC article.
-
Novel Strategies for Spatiotemporal and Controlled BMP-2 Delivery in Bone Tissue Engineering.Cell Transplant. 2024 Jan-Dec;33:9636897241276733. doi: 10.1177/09636897241276733. Cell Transplant. 2024. PMID: 39305020 Free PMC article. Review.
-
Revolutionizing bone defect healing: the power of mesenchymal stem cells as seeds.Front Bioeng Biotechnol. 2024 Oct 21;12:1421674. doi: 10.3389/fbioe.2024.1421674. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 39497791 Free PMC article. Review.
Cited by
-
Identification of core genes and prediction of miRNAs associated with osteoporosis using a bioinformatics approach.Oncol Lett. 2019 Jan;17(1):468-481. doi: 10.3892/ol.2018.9508. Epub 2018 Sep 26. Oncol Lett. 2019. PMID: 30655789 Free PMC article.
-
Functionalized Annealed Microgels for Spatial Control of Osteogenic and Chondrogenic Differentiation.Adv Funct Mater. 2024 Jul 24;34(30):2311017. doi: 10.1002/adfm.202311017. Epub 2024 Jun 10. Adv Funct Mater. 2024. PMID: 40800235 Free PMC article.
-
BMP2-modified injectable hydrogel for osteogenic differentiation of human periodontal ligament stem cells.Sci Rep. 2017 Jul 26;7(1):6603. doi: 10.1038/s41598-017-06911-8. Sci Rep. 2017. PMID: 28747761 Free PMC article.
-
Two Hits for Bone Regeneration in Aged Patients: Vertebral Bone Marrow Clot as a Biological Scaffold and Powerful Source of Mesenchymal Stem Cells.Front Bioeng Biotechnol. 2022 Jan 18;9:807679. doi: 10.3389/fbioe.2021.807679. eCollection 2021. Front Bioeng Biotechnol. 2022. PMID: 35118056 Free PMC article.
-
Functional Hydrogels With Tunable Structures and Properties for Tissue Engineering Applications.Front Chem. 2018 Oct 22;6:499. doi: 10.3389/fchem.2018.00499. eCollection 2018. Front Chem. 2018. PMID: 30406081 Free PMC article. Review.
References
-
- Calori GM, Colombo M, Mazza EL, Mazzola S, Malagoli E, Mineo GV. Incidence of donor site morbidity following harvesting from iliac crest or RIA graft. Injury. 2014;45(Suppl 6):S116–S120. - PubMed
-
- Gomez-Barrena E, Rosset P, Lozano D, Stanovici J, Ermthaller C, Gerbhard F. Bone fracture healing: cell therapy in delayed unions and nonunions. Bone. 2015;70:93–101. - PubMed
-
- Wen C, Kang H, Shih YR, Hwang Y, Varghese S. In vivo comparison of biomineralized scaffold-directed osteogenic differentiation of human embryonic and mesenchymal stem cells. Drug Deliv Transl Res. 2016;6:121–131. - PubMed
-
- Song S, Kim EJ, Bahney CS, Miclau T, Marcucio R, Roy S. The synergistic effect of micro-topography and biochemical culture environment to promote angiogenesis and osteogenic differentiation of human mesenchymal stem cells. Acta Biomater. 2015;18:100–111. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

