Residues K465 and G467 within the Cytoplasmic Domain of GP2 Play a Critical Role in the Persistence of Lymphocytic Choriomeningitis Virus in Mice
- PMID: 27581982
- PMCID: PMC5105648
- DOI: 10.1128/JVI.01303-16
Residues K465 and G467 within the Cytoplasmic Domain of GP2 Play a Critical Role in the Persistence of Lymphocytic Choriomeningitis Virus in Mice
Abstract
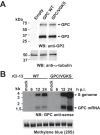
Several arenaviruses, chiefly Lassa virus (LASV), cause hemorrhagic fever disease in humans and pose serious public health concerns in their regions of endemicity. Moreover, mounting evidence indicates that the worldwide-distributed prototypic arenavirus, lymphocytic choriomeningitis virus (LCMV), is a neglected human pathogen of clinical significance. We have documented that a recombinant LCMV containing the glycoprotein (GPC) gene of LASV within the backbone of the immunosuppressive clone 13 (Cl-13) variant of the Armstrong strain of LCMV (rCl-13/LASV-GPC) exhibited Cl-13-like growth properties in cultured cells, but in contrast to Cl-13, rCl-13/LASV-GPC was unable to establish persistence in immunocompetent adult mice, which prevented its use for some in vivo experiments. Recently, V459K and K461G mutations within the GP2 cytoplasmic domain (CD) of rCl-13/LASV-GPC were shown to increase rCl-13/LASV-GPC infectivity in mice. Here, we generated rCl-13(GPC/VGKS) by introducing the corresponding revertant mutations K465V and G467K within GP2 of rCl-13 and we show that rCl-13(GPC/VGKS) was unable to persist in mice. K465V and G467K mutations did not affect GPC processing, virus RNA replication, or gene expression. In addition, rCl-13(GPC/VGKS) grew to high titers in cultured cell lines and in immunodeficient mice. Further analysis revealed that rCl-13(GPC/VGKS) infected fewer splenic plasmacytoid dendritic cells than rCl-13, yet the two viruses induced similar type I interferon responses in mice. Our findings have identified novel viral determinants of Cl-13 persistence and also revealed that virus GPC-host interactions yet to be elucidated critically contribute to Cl-13 persistence.
Importance: The prototypic arenavirus, lymphocytic choriomeningitis virus (LCMV), provides investigators with a superb experimental model system to investigate virus-host interactions. The Armstrong strain (ARM) of LCMV causes an acute infection, whereas its derivative, clone 13 (Cl-13), causes a persistent infection. Mutations F260L and K1079Q within GP1 and L polymerase, respectively, have been shown to play critical roles in Cl-13's ability to persist in mice. However, there is an overall lack of knowledge about other viral determinants required for Cl-13's persistence. Here, we report that mutations K465V and G467K within the cytoplasmic domain of Cl-13 GP2 resulted in a virus, rCl-13(GPC/VGKS), that failed to persist in mice despite exhibiting Cl-13 wild-type-like fitness in cultured cells and immunocompromised mice. This finding has uncovered novel viral determinants of viral persistence, and a detailed characterization of rCl-13(GPC/VGKS) can provide novel insights into the mechanisms underlying persistent viral infection.
Copyright © 2016, American Society for Microbiology. All Rights Reserved.
Figures


Similar articles
-
Evolution of recombinant lymphocytic choriomeningitis virus/Lassa virus in vivo highlights the importance of the GPC cytosolic tail in viral fitness.J Virol. 2014 Aug;88(15):8340-8. doi: 10.1128/JVI.00236-14. Epub 2014 May 14. J Virol. 2014. PMID: 24829355 Free PMC article.
-
A multivalent vaccination strategy for the prevention of Old World arenavirus infection in humans.J Virol. 2010 Oct;84(19):9947-56. doi: 10.1128/JVI.00672-10. Epub 2010 Jul 28. J Virol. 2010. PMID: 20668086 Free PMC article.
-
A Highly Conserved Leucine in Mammarenavirus Matrix Z Protein Is Required for Z Interaction with the Virus L Polymerase and Z Stability in Cells Harboring an Active Viral Ribonucleoprotein.J Virol. 2018 May 14;92(11):e02256-17. doi: 10.1128/JVI.02256-17. Print 2018 Jun 1. J Virol. 2018. PMID: 29593035 Free PMC article.
-
Lassa virus glycoprotein complex review: insights into its unique fusion machinery.Biosci Rep. 2022 Feb 25;42(2):BSR20211930. doi: 10.1042/BSR20211930. Biosci Rep. 2022. PMID: 35088070 Free PMC article. Review.
-
Arenavirus reverse genetics: new approaches for the investigation of arenavirus biology and development of antiviral strategies.Virology. 2011 Mar 15;411(2):416-25. doi: 10.1016/j.virol.2011.01.013. Epub 2011 Feb 15. Virology. 2011. PMID: 21324503 Free PMC article. Review.
Cited by
-
An mRNA-LNP-based Lassa virus vaccine induces protective immunity in mice.J Virol. 2024 Jun 13;98(6):e0057824. doi: 10.1128/jvi.00578-24. Epub 2024 May 20. J Virol. 2024. PMID: 38767352 Free PMC article.
-
Biological Characterization of Conserved Residues within the Cytoplasmic Tail of the Pichinde Arenaviral Glycoprotein Subunit 2 (GP2).J Virol. 2019 Oct 29;93(22):e01277-19. doi: 10.1128/JVI.01277-19. Print 2019 Nov 15. J Virol. 2019. PMID: 31462569 Free PMC article.
References
-
- Buchmeier MJ, Peters CJ, de la Torre JC. 2007. Arenaviridae: the viruses and their replication, p 1791–1851. In Knipe DM, Howley PM, Griffin DE, Lamb RA, Martin MA, Roizman B, Straus SE (ed), Fields virology, 5th ed, vol 2 Lippincott Williams & Wilkins, Philadelphia, PA.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

