Development and Validation of a Histological Method to Measure Microvessel Density in Whole-Slide Images of Cancer Tissue
- PMID: 27583442
- PMCID: PMC5008750
- DOI: 10.1371/journal.pone.0161496
Development and Validation of a Histological Method to Measure Microvessel Density in Whole-Slide Images of Cancer Tissue
Abstract
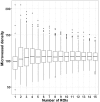
Despite all efforts made to develop predictive biomarkers for antiangiogenic therapies, no unambiguous markers have been identified so far. This is due to among others the lack of standardized tests. This study presents an improved microvessel density quantification method in tumor tissue based on stereological principles and using whole-slide images. Vessels in tissue sections of different cancer types were stained for CD31 by an automated and validated immunohistochemical staining method. The stained slides were digitized with a digital slide scanner. Systematic, uniform, random sampling of the regions of interest on the whole-slide images was performed semi-automatically with the previously published applications AutoTag and AutoSnap. Subsequently, an unbiased counting grid was combined with the images generated with these scripts. Up to six independent observers counted microvessels in up to four cancer types: colorectal carcinoma, glioblastoma multiforme, ovarian carcinoma and renal cell carcinoma. At first, inter-observer variability was found to be unacceptable. However, after a series of consensus training sessions and interim statistical analysis, counting rules were modified and inter-observer concordance improved considerably. Every CD31-positive object was counted, with exclusion of suspected CD31-positive monocytes, macrophages and tumor cells. Furthermore, if interconnected, stained objects were considered a single vessel. Ten regions of interest were sufficient for accurate microvessel density measurements. Intra-observer and inter-observer variability were low (intraclass correlation coefficient > 0.7) if the observers were adequately trained.
Conflict of interest statement
The authors have read the journal's policy and the authors of this manuscript have the following competing interests: Koen Marien, Valerie Croons, Yannick Waumans, Ellen Sluydts, Stefanie De Schepper, and Luc Andries are employees of HistoGeneX NV (Antwerp, Belgium). Mark Kockx is CEO of HistoGeneX NV, which carries out immunohistochemical testing for pharmaceutical companies as part of (pre-)clinical studies that evaluate new anticancer drugs. Mark Kockx and Wim Waelput are consulting pathologists to Pathomation BVBA. The authors have no other relevant affiliations or financial involvements with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. This does not alter the authors' adherence to PLOS ONE policies on sharing data and materials.
Figures




References
-
- Weidner N. Current pathologic methods for measuring intratumoral microvessel density within breast carcinoma and other solid tumors. Breast Cancer Res Treat. 1995;36: 169–80. - PubMed
-
- Vermeulen PB, Gasparini G, Fox SB, Toi M, Martin L, McCulloch P, et al. Quantification of angiogenesis in solid human tumours: an international consensus on the methodology and criteria of evaluation. Eur J Cancer. 1996;32A: 2474–84. - PubMed
-
- Vermeulen PB, Gasparini G, Fox SB, Colpaert C, Marson LP, Gion M, et al. Second international consensus on the methodology and criteria of evaluation of angiogenesis quantification in solid human tumours. Eur J Cancer. 2002;38: 1564–79. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

