TIMELESS Suppresses the Accumulation of Aberrant CDC45·MCM2-7·GINS Replicative Helicase Complexes on Human Chromatin
- PMID: 27587400
- PMCID: PMC5077192
- DOI: 10.1074/jbc.M116.719963
TIMELESS Suppresses the Accumulation of Aberrant CDC45·MCM2-7·GINS Replicative Helicase Complexes on Human Chromatin
Abstract
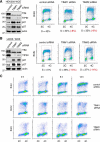
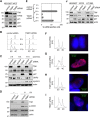
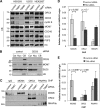
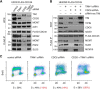
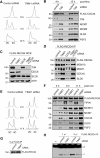
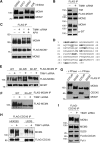
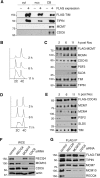
The replication licensing factor CDC6 recruits the MCM2-7 replicative helicase to the replication origin, where MCM2-7 is activated to initiate DNA replication. MCM2-7 is activated by both the CDC7-Dbf4 kinase and cyclin-dependent kinase and via interactions with CDC45 and go-ichi-ni-san complex (GINS) to form the CDC45·MCM2-7·GINS (CMG) helicase complex. TIMELESS (TIM) is important for the subsequent coupling of CMG activity to DNA polymerases for efficient DNA synthesis. However, the mechanism by which TIM regulates CMG activity for proper replication fork progression remains unclear. Here we show that TIM interacts with MCM2-7 prior to the initiation of DNA replication. TIM depletion in various human cell lines results in the accumulation of aberrant CMG helicase complexes on chromatin. Importantly, the presence of these abnormal CMG helicase complexes is not restricted to cells undergoing DNA synthesis. Furthermore, even though these aberrant CMG complexes interact with the DNA polymerases on human chromatin, these complexes are not phosphorylated properly by cyclin-dependent kinase/CDC7-Dbf4 kinase and exhibit reduced DNA unwinding activity. This phenomenon coincides with a significant accumulation of the p27 and p21 replication inhibitors, reduced chromatin association of CDC6 and cyclin E, and a delay in S phase entry. Our results provide the first evidence that TIM is required for the correct chromatin association of the CMG complex to allow efficient DNA replication.
Keywords: DNA binding protein; DNA helicase; DNA replication; cell cycle; cyclin-dependent kinase (CDK); protein complex.
© 2016 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

Similar articles
-
DDK regulates replication initiation by controlling the multiplicity of Cdc45-GINS binding to Mcm2-7.Elife. 2021 Feb 22;10:e65471. doi: 10.7554/eLife.65471. Elife. 2021. PMID: 33616038 Free PMC article.
-
The replication initiation protein Sld2 regulates helicase assembly.J Biol Chem. 2014 Jan 24;289(4):1948-59. doi: 10.1074/jbc.M113.532085. Epub 2013 Dec 4. J Biol Chem. 2014. PMID: 24307213 Free PMC article.
-
The Dbf4-Cdc7 kinase promotes Mcm2-7 ring opening to allow for single-stranded DNA extrusion and helicase assembly.J Biol Chem. 2015 Jan 9;290(2):1210-21. doi: 10.1074/jbc.M114.608232. Epub 2014 Dec 3. J Biol Chem. 2015. Retraction in: J Biol Chem. 2017 Jun 16;292(24):10320. doi: 10.1074/jbc.A114.608232. PMID: 25471369 Free PMC article. Retracted.
-
Helicase activation and establishment of replication forks at chromosomal origins of replication.Cold Spring Harb Perspect Biol. 2013 Dec 1;5(12):a010371. doi: 10.1101/cshperspect.a010371. Cold Spring Harb Perspect Biol. 2013. PMID: 23881938 Free PMC article. Review.
-
Behavior of replication origins in Eukaryota - spatio-temporal dynamics of licensing and firing.Cell Cycle. 2015;14(14):2251-64. doi: 10.1080/15384101.2015.1056421. Epub 2015 Jun 1. Cell Cycle. 2015. PMID: 26030591 Free PMC article. Review.
Cited by
-
DNA replication initiation factor RECQ4 possesses a role in antagonizing DNA replication initiation.Nat Commun. 2023 Mar 4;14(1):1233. doi: 10.1038/s41467-023-36968-1. Nat Commun. 2023. PMID: 36871012 Free PMC article.
-
Mechanism for local attenuation of DNA replication at double-strand breaks.Nature. 2025 Mar;639(8056):1084-1092. doi: 10.1038/s41586-024-08557-9. Epub 2025 Feb 19. Nature. 2025. PMID: 39972127
-
Pathogenic mutations reveal a role of RECQ4 in mitochondrial RNA:DNA hybrid formation and resolution.Sci Rep. 2020 Oct 12;10(1):17033. doi: 10.1038/s41598-020-74095-9. Sci Rep. 2020. PMID: 33046774 Free PMC article.
-
Molecular Mechanisms of the RECQ4 Pathogenic Mutations.Front Mol Biosci. 2021 Nov 18;8:791194. doi: 10.3389/fmolb.2021.791194. eCollection 2021. Front Mol Biosci. 2021. PMID: 34869606 Free PMC article. Review.
-
CDC45 modulates MCM7 expression and inhibits cell proliferation by suppressing the PI3K/AKT pathway in acute myeloid leukemia.Am J Transl Res. 2021 Sep 15;13(9):10218-10232. eCollection 2021. Am J Transl Res. 2021. PMID: 34650692 Free PMC article.
References
-
- Edwards M. C., Tutter A. V., Cvetic C., Gilbert C. H., Prokhorova T. A., and Walter J. C. (2002) MCM2-7 complexes bind chromatin in a distributed pattern surrounding the origin recognition complex in Xenopus egg extracts. J. Biol. Chem. 277, 33049–33057 - PubMed
-
- Kim J., and Kipreos E. T. (2008) Control of the Cdc6 replication licensing factor in metazoa: the role of nuclear export and the CUL4 ubiquitin ligase. Cell Cycle 7, 146–150 - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

