In Vitro Testing of Biomaterials for Neural Repair: Focus on Cellular Systems and High-Content Analysis
- PMID: 27588220
- PMCID: PMC4991583
- DOI: 10.1089/biores.2016.0025
In Vitro Testing of Biomaterials for Neural Repair: Focus on Cellular Systems and High-Content Analysis
Abstract
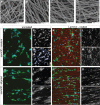
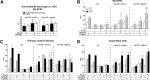
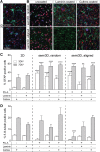
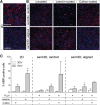
Biomimetic materials are designed to stimulate specific cellular responses at the molecular level. To improve the soundness of in vitro testing of the biological impact of new materials, appropriate cell systems and technologies must be standardized also taking regulatory issues into consideration. In this study, the biological and molecular effects of different scaffolds on three neural systems, that is, the neural cell line SH-SY5Y, primary cortical neurons, and neural stem cells, were compared. The effect of poly(L-lactic acid) scaffolds having different surface geometry (conventional two-dimensional seeding flat surface, random or aligned fibers as semi3D structure) and chemical functionalization (laminin or ECM extract) were studied. The endpoints were defined for efficacy (i.e., neural differentiation and neurite elongation) and for safety (i.e., cell death/survival) using high-content analysis. It is demonstrated that (i) the definition of the biological properties of biomaterials is profoundly influenced by the test system used; (ii) the definition of the in vitro safety profile of biomaterials for neural repair is also influenced by the test system; (iii) cell-based high-content screening may well be successfully used to characterize both the efficacy and safety of novel biomaterials, thus speeding up and improving the soundness of this critical step in material science having medical applications.
Keywords: Good Laboratory Practice guidelines; neural cell lines; neural primary culture; neural stem cells high-content analysis; poly(lactic acid).
Figures

Similar articles
-
Culture of pyramidal neural precursors, neural stem cells, and fibroblasts on various biomaterials.J Biomater Sci Polym Ed. 2018 Dec;29(17):2168-2186. doi: 10.1080/09205063.2018.1528520. Epub 2018 Dec 14. J Biomater Sci Polym Ed. 2018. PMID: 30261158
-
Electrospinning of nano/micro scale poly(L-lactic acid) aligned fibers and their potential in neural tissue engineering.Biomaterials. 2005 May;26(15):2603-10. doi: 10.1016/j.biomaterials.2004.06.051. Biomaterials. 2005. PMID: 15585263
-
Patterned and functionalized nanofiber scaffolds in three-dimensional hydrogel constructs enhance neurite outgrowth and directional control.J Neural Eng. 2014 Dec;11(6):066009. doi: 10.1088/1741-2560/11/6/066009. Epub 2014 Oct 31. J Neural Eng. 2014. PMID: 25358624
-
Biomimetic Materials and Their Utility in Modeling the 3-Dimensional Neural Environment.iScience. 2020 Jan 24;23(1):100788. doi: 10.1016/j.isci.2019.100788. Epub 2019 Dec 19. iScience. 2020. PMID: 31954980 Free PMC article. Review.
-
Scaffolds for peripheral nerve repair and reconstruction.Exp Neurol. 2019 Sep;319:112761. doi: 10.1016/j.expneurol.2018.05.016. Epub 2018 Jun 2. Exp Neurol. 2019. PMID: 29772248 Review.
Cited by
-
Oligodendrocytes in a dish for the drug discovery pipeline: the risk of oversimplification.Neural Regen Res. 2021 Feb;16(2):291-293. doi: 10.4103/1673-5374.290888. Neural Regen Res. 2021. PMID: 32859780 Free PMC article. No abstract available.
-
Intra-individual variability in the neuroprotective and promyelinating properties of conditioned culture medium obtained from human adipose mesenchymal stromal cells.Stem Cell Res Ther. 2023 May 11;14(1):128. doi: 10.1186/s13287-023-03344-1. Stem Cell Res Ther. 2023. PMID: 37170115 Free PMC article.
-
The Influence of Pathological Extracellular Matrix on the Biological Properties of Stem Cells: Possible Hints for Cell Transplantation Therapies in Spinal Cord Injury.Int J Mol Sci. 2025 Apr 23;26(9):3969. doi: 10.3390/ijms26093969. Int J Mol Sci. 2025. PMID: 40362209 Free PMC article.
-
Effect of Electrical Stimulation on PC12 Cells Cultured in Different Hydrogels: Basis for the Development of Biomaterials in Peripheral Nerve Tissue Engineering.Pharmaceutics. 2023 Dec 12;15(12):2760. doi: 10.3390/pharmaceutics15122760. Pharmaceutics. 2023. PMID: 38140099 Free PMC article.
-
A Novel Three-Dimensional Culture Device Favors a Myelinating Morphology of Neural Stem Cell-Derived Oligodendrocytes.Front Cell Dev Biol. 2021 Oct 1;9:759982. doi: 10.3389/fcell.2021.759982. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34660610 Free PMC article.
References
-
- Cao H, Liu T, Chew SY. The application of nanofibrous scaffolds in neural tissue engineering. Adv Drug Deliv Rev. 2009;61:1055–1064 - PubMed
-
- Russell AJ. Standardized experimental procedures in tissue engineering: cure or curse. Tissue Eng. 2005;11:vii–ix - PubMed
-
- Amato S, Ezzell B. Regulatory Affairs for Biomaterials and Medical Devices. WP Woodhead Publising, Sawston, Cambridge, 2015
LinkOut - more resources
Full Text Sources
Other Literature Sources
