Melanin-concentrating hormone neurons specifically promote rapid eye movement sleep in mice
- PMID: 27595887
- PMCID: PMC5056843
- DOI: 10.1016/j.neuroscience.2016.08.046
Melanin-concentrating hormone neurons specifically promote rapid eye movement sleep in mice
Abstract
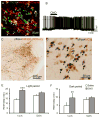
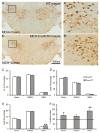
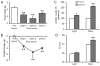
Currently available evidence indicates that neurons containing melanin-concentrating hormone (MCH) in the lateral hypothalamus are critical modulators of sleep-wakefulness, but their precise role in this function is not clear. Studies employing optogenetic stimulation of MCH neurons have yielded inconsistent results, presumably due to differences in the optogenetic stimulation protocols, which do not approximate normal patterns of cell firing. In order to resolve this discrepancy, we (1) selectively activated the MCH neurons using a chemogenetic approach (Cre-dependent hM3Dq expression) and (2) selectively destroyed MCH neurons using a genetically targeted diphtheria toxin deletion method, and studied the changes in sleep-wake in mice. Our results indicate that selective activation of MCH neurons causes specific increases in rapid eye movement (REM) sleep without altering wake or non-REM (NREM) sleep. On the other hand, selective deletions of MCH neurons altered the diurnal rhythm of wake and REM sleep without altering their total amounts. These results indicate that activation of MCH neurons primarily drives REM sleep and their presence may be necessary for normal expression of diurnal variation of REM sleep and wake.
Keywords: chemogenetics; conditional deletion; feeding; lateral hypothalamus; paradoxical sleep; sleep–wake.
Copyright © 2016 IBRO. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
Similar articles
-
Melanin-concentrating hormone neurons contribute to dysregulation of rapid eye movement sleep in narcolepsy.Neurobiol Dis. 2018 Dec;120:12-20. doi: 10.1016/j.nbd.2018.08.012. Epub 2018 Aug 24. Neurobiol Dis. 2018. PMID: 30149182 Free PMC article.
-
Optogenetic activation of melanin-concentrating hormone neurons increases non-rapid eye movement and rapid eye movement sleep during the night in rats.Eur J Neurosci. 2016 Nov;44(10):2846-2857. doi: 10.1111/ejn.13410. Epub 2016 Oct 16. Eur J Neurosci. 2016. PMID: 27657541 Free PMC article.
-
Melanin-concentrating hormone neurons promote rapid eye movement sleep independent of glutamate release.Brain Struct Funct. 2019 Jan;224(1):99-110. doi: 10.1007/s00429-018-1766-2. Epub 2018 Oct 3. Brain Struct Funct. 2019. PMID: 30284033 Free PMC article.
-
Role of the melanin-concentrating hormone neuropeptide in sleep regulation.Peptides. 2009 Nov;30(11):2052-9. doi: 10.1016/j.peptides.2009.07.022. Epub 2009 Aug 4. Peptides. 2009. PMID: 19660508 Review.
-
Sleep-Wake Control by Melanin-Concentrating Hormone (MCH) Neurons: a Review of Recent Findings.Curr Neurol Neurosci Rep. 2020 Oct 2;20(12):55. doi: 10.1007/s11910-020-01075-x. Curr Neurol Neurosci Rep. 2020. PMID: 33006677 Free PMC article. Review.
Cited by
-
Whole-Brain Neural Connectivity to Lateral Pontine Tegmentum GABAergic Neurons in Mice.Front Neurosci. 2019 Apr 24;13:375. doi: 10.3389/fnins.2019.00375. eCollection 2019. Front Neurosci. 2019. PMID: 31068780 Free PMC article.
-
Role of Hypothalamus in Acupuncture's Effects.Brain Sci. 2025 Jan 15;15(1):72. doi: 10.3390/brainsci15010072. Brain Sci. 2025. PMID: 39851439 Free PMC article. Review.
-
Melanin-concentrating hormone neurons contribute to dysregulation of rapid eye movement sleep in narcolepsy.Neurobiol Dis. 2018 Dec;120:12-20. doi: 10.1016/j.nbd.2018.08.012. Epub 2018 Aug 24. Neurobiol Dis. 2018. PMID: 30149182 Free PMC article.
-
Characterization of Hypothalamic MCH Neuron Development in a 3D Differentiation System of Mouse Embryonic Stem Cells.eNeuro. 2022 Apr 26;9(2):ENEURO.0442-21.2022. doi: 10.1523/ENEURO.0442-21.2022. Print 2022 Mar-Apr. eNeuro. 2022. PMID: 35437265 Free PMC article.
-
Global dissociation of the posterior amygdala from the rest of the brain during REM sleep.Commun Biol. 2022 Nov 28;5(1):1306. doi: 10.1038/s42003-022-04257-0. Commun Biol. 2022. PMID: 36443640 Free PMC article.
References
-
- Ahnaou A, Drinkenburg WH, Bouwknecht JA, Alcazar J, Steckler T, Dautzenberg FM. Blocking melanin-concentrating hormone MCH1 receptor affects rat sleep-wake architecture. Eur J Pharmacol. 2008;579:177–188. - PubMed
-
- Astrand A, Bohlooly YM, Larsdotter S, Mahlapuu M, Andersen H, Tornell J, Ohlsson C, Snaith M, Morgan DG. Mice lacking melanin-concentrating hormone receptor 1 demonstrate increased heart rate associated with altered autonomic activity. Am J Physiol Regul Integr Comp Physiol. 2004;287:R749–758. - PubMed
MeSH terms
Substances
Grants and funding
- R21 NS097922/NS/NINDS NIH HHS/United States
- R01 DK089044/DK/NIDDK NIH HHS/United States
- R01 DK096010/DK/NIDDK NIH HHS/United States
- R37 DK053477/DK/NIDDK NIH HHS/United States
- K01 DK094943/DK/NIDDK NIH HHS/United States
- R01 DK071051/DK/NIDDK NIH HHS/United States
- R01 NS062727/NS/NINDS NIH HHS/United States
- P30 DK046200/DK/NIDDK NIH HHS/United States
- R01 NS085477/NS/NINDS NIH HHS/United States
- R21 NS074205/NS/NINDS NIH HHS/United States
- R01 NS091126/NS/NINDS NIH HHS/United States
- P01 AG009975/AG/NIA NIH HHS/United States
- R01 DK108797/DK/NIDDK NIH HHS/United States
- P30 DK057521/DK/NIDDK NIH HHS/United States
- R01 NS088482/NS/NINDS NIH HHS/United States
- R01 DK075632/DK/NIDDK NIH HHS/United States
- R01 NS061841/NS/NINDS NIH HHS/United States
- P01 HL095491/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

