Synaptic Mitochondria Sustain More Damage than Non-Synaptic Mitochondria after Traumatic Brain Injury and Are Protected by Cyclosporine A
- PMID: 27596283
- PMCID: PMC5385586
- DOI: 10.1089/neu.2016.4628
Synaptic Mitochondria Sustain More Damage than Non-Synaptic Mitochondria after Traumatic Brain Injury and Are Protected by Cyclosporine A
Abstract
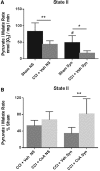
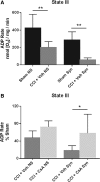
Currently, there are no Food and Drug Administration (FDA)-approved pharmacotherapies for the treatment of those with traumatic brain injury (TBI). As central mediators of the secondary injury cascade, mitochondria are promising therapeutic targets for prevention of cellular death and dysfunction after TBI. One of the most promising and extensively studied mitochondrial targeted TBI therapies is inhibition of the mitochondrial permeability transition pore (mPTP) by the FDA-approved drug, cyclosporine A (CsA). A number of studies have evaluated the effects of CsA on total brain mitochondria after TBI; however, no study has investigated the effects of CsA on isolated synaptic and non-synaptic mitochondria. Synaptic mitochondria are considered essential for proper neurotransmission and synaptic plasticity, and their dysfunction has been implicated in neurodegeneration. Synaptic and non-synaptic mitochondria have heterogeneous characteristics, but their heterogeneity can be masked in total mitochondrial (synaptic and non-synaptic) preparations. Therefore, it is essential that mitochondria targeted pharmacotherapies, such as CsA, be evaluated in both populations. This is the first study to examine the effects of CsA on isolated synaptic and non-synaptic mitochondria after experimental TBI. We conclude that synaptic mitochondria sustain more damage than non-synaptic mitochondria 24 h after severe controlled cortical impact injury (CCI), and that intraperitoneal administration of CsA (20 mg/kg) 15 min after injury improves synaptic and non-synaptic respiration, with a significant improvement being seen in the more severely impaired synaptic population. As such, CsA remains a promising neuroprotective candidate for the treatment of those with TBI.
Keywords: cyclosporine A; mitochondria; neuroprotection; synaptic and non-synaptic; traumatic brain injury.
Conflict of interest statement
No competing financial interests exist.
Figures




References
-
- Thurman D.J., Alverson C., Dunn K.A., Guerrero J., and Sniezek J.E. (1999). Traumatic brain injury in the United States: a public health perspective. J. Head Trauma Rehabil. 14, 602–615 - PubMed
-
- Finkelstein E.A, Corso P.S., and Miller T.R. (2006). The Incidence and Economic Burden of Injuries in the United States. Oxford University Press: New York
-
- Coronado V.G., McGuire L.C., Faul M.F., Sugerman D.E., and Pearson W.S. (2012). Traumatic brain injury epidemiology and public health issues. in: Brain Injury Medicine: Principles and Practice. Zasler N.D., Katz D.I., and Zafonte R.D. (eds). Demos Medical Publishing: New York, pps. 84–100
-
- Maas A.I., Stocchetti N., and Bullock R. (2008). Moderate and severe traumatic brain injury in adults. Lancet Neurol. 7, 728–741 - PubMed
-
- Faden A.I., Demediuk P., Panter S.S., and Vink R. (1989). The role of excitatory amino acids and NMDA receptors in traumatic brain injury. Science 244, 798–800 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

