Comparative effect of immature neuronal or glial cell transplantation on motor functional recovery following experimental traumatic brain injury in rats
- PMID: 27602084
- PMCID: PMC4998226
- DOI: 10.3892/etm.2016.3527
Comparative effect of immature neuronal or glial cell transplantation on motor functional recovery following experimental traumatic brain injury in rats
Abstract
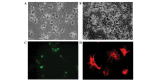
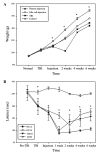
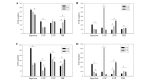
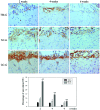
The present study evaluated the comparative effect of stereotaxically transplanted immature neuronal or glial cells in brain on motor functional recovery and cytokine expression after cold-induced traumatic brain injury (TBI) in adult rats. A total of 60 rats were divided into four groups (n=15/group): Sham group; TBI only group; TBI plus neuronal cells-transplanted group (NC-G); and TBI plus glial cells-transplanted group (GC-G). Cortical lesions were induced by a touching metal stamp, frozen with liquid nitrogen, to the dura mater over the motor cortex of adult rats. Neuronal and glial cells were isolated from rat embryonic and newborn cortices, respectively, and cultured in culture flasks. Rats received neurons or glia grafts (~1×106 cells) 5 days after TBI was induced. Motor functional evaluation was performed with the rotarod test prior to and following glial and neural cell grafts. Five rats from each group were sacrificed at 2, 4 and 6 weeks post-cell transplantation. Immunofluorescence staining was performed on brain section to identify the transplanted neuronal or glial cells using neural and astrocytic markers. The expression levels of cytokines, including transforming growth factor-β, glial cell-derived neurotrophic factor and vascular endothelial growth factor, which have key roles in the proliferation, differentiation and survival of neural cells, were analyzed by immunohistochemistry and western blotting. A localized cortical lesion was evoked in all injured rats, resulting in significant motor deficits. Transplanted cells successfully migrated and survived in the injured brain lesion, and the expression of neuronal and astrocyte markers were detected in the NC-G and GC-G groups, respectively. Rats in the NC-G and GC-G cell-transplanted groups exhibited significant motor functional recovery and reduced histopathologic lesions, as compared with the TBI-G rats that did not receive neural cells (P<0.05, respectively). Furthermore, GC-G treatment induced significantly improved motor functional recovery, as compared with the NC-G group (P<0.05). Increased cytokine expression levels were detected in the NC-G and GC-G groups, as compared with the TBI-G; however, no differences were found between the two groups. These data suggested that transplanted immature neural cells may promote the survival of neural cells in cortical lesion and motor functional recovery. Furthermore, transplanted glial cells may be used as an effective therapeutic tool for TBI patients with abnormalities in motor functional recovery and cytokine expression.
Keywords: glia; motor functional recovery; neuron; transplantation; traumatic brain injury.
Figures



References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
