No diurnal variation of classical and candidate biomarkers of Alzheimer's disease in CSF
- PMID: 27605218
- PMCID: PMC5013624
- DOI: 10.1186/s13024-016-0130-3
No diurnal variation of classical and candidate biomarkers of Alzheimer's disease in CSF
Abstract
Background: Cerebrospinal fluid (CSF) biomarkers have gained increasing importance in the diagnostic work-up of Alzheimer's disease (AD). The core CSF biomarkers related to AD pathology (Aβ42, t-tau and p-tau) are currently used in CSF diagnostics, while candidate markers of amyloid metabolism (Aβ38, Aβ40, sAPPα, sAPPβ), synaptic loss (neurogranin), neuroinflammation (YKL-40), neuronal damage (VILIP-1) and genetic risk (apolipoprotein E) are undergoing evaluation. Diurnal fluctuation in the concentration of CSF biomarkers has been reported and may represent a preanalytical confounding factor in the laboratory diagnosis of AD. The aim of the present study was to investigate the diurnal variability of classical and candidate CSF biomarkers in a cohort of neurosurgical patients carrying a CSF drainage.
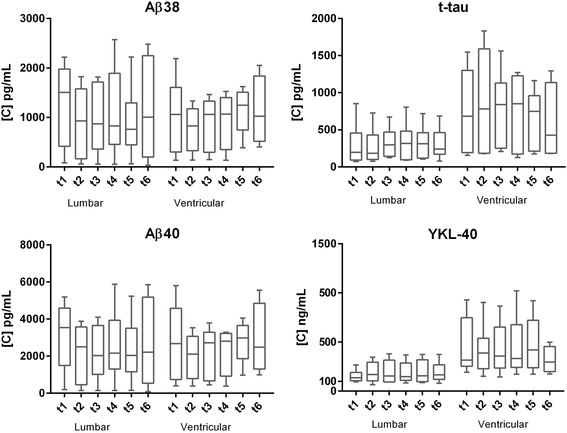
Method: Samples were collected from a cohort of 13 neurosurgical patients from either ventricular (n = 6) or lumbar (n = 7) CSF drainage at six time points during the day, 1-7 days following the neurosurgical intervention. Concentrations of the core biomarkers were determined by immunoassays.
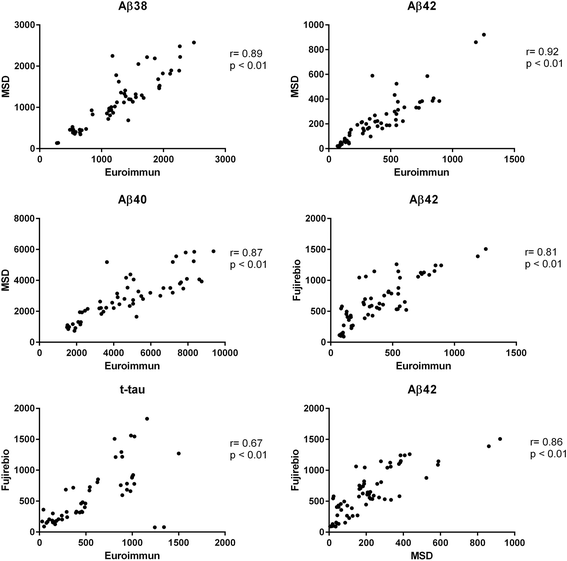
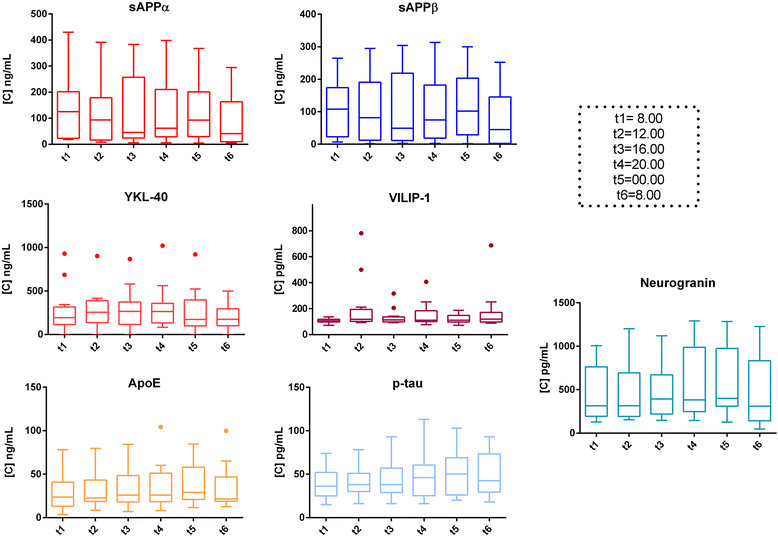
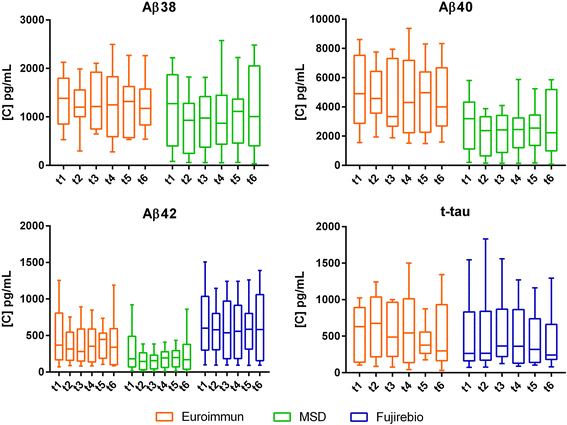
Results: Although absolute values largely varied among subjects, none of the biomarkers showed significant diurnal variation. Site of drainage (lumbar vs. ventricular) did not influence this result. The different immunoassays used for tau and Aβ markers provided similar results.
Conclusion: Time of day at CSF collection does not ultimately affect the concentration levels of classical and candidate AD biomarkers. Similar trends were found when using different immunoassays, thus corroborating the consistency of the data.
Figures
Comment in
-
Diurnal oscillation of CSF Aβ and other AD biomarkers.Mol Neurodegener. 2017 May 8;12(1):36. doi: 10.1186/s13024-017-0161-4. Mol Neurodegener. 2017. PMID: 28478762 Free PMC article.
Similar articles
-
Effects of time of the day at sampling on CSF and plasma levels of Alzheimer' disease biomarkers.Alzheimers Res Ther. 2024 Jun 22;16(1):132. doi: 10.1186/s13195-024-01503-x. Alzheimers Res Ther. 2024. PMID: 38909218 Free PMC article.
-
Apolipoprotein E genotype and the diagnostic accuracy of cerebrospinal fluid biomarkers for Alzheimer disease.JAMA Psychiatry. 2014 Oct;71(10):1183-91. doi: 10.1001/jamapsychiatry.2014.1060. JAMA Psychiatry. 2014. PMID: 25162367
-
Additional use of Aβ₄₂/Aβ₄₀ ratio with cerebrospinal fluid biomarkers P-tau and Aβ₄₂ increases the level of evidence of Alzheimer's disease pathophysiological process in routine practice.J Alzheimers Dis. 2014;41(2):377-86. doi: 10.3233/JAD-131838. J Alzheimers Dis. 2014. PMID: 24614902
-
Simultaneous analysis of cerebrospinal fluid biomarkers using microsphere-based xMAP multiplex technology for early detection of Alzheimer's disease.Methods. 2012 Apr;56(4):484-93. doi: 10.1016/j.ymeth.2012.03.023. Epub 2012 Apr 6. Methods. 2012. PMID: 22503777 Review.
-
Alzheimer's disease--Recent biomarker developments in relation to updated diagnostic criteria.Clin Chim Acta. 2015 Sep 20;449:3-8. doi: 10.1016/j.cca.2015.01.041. Epub 2015 Feb 7. Clin Chim Acta. 2015. PMID: 25668231 Review.
Cited by
-
Effects of time of the day at sampling on CSF and plasma levels of Alzheimer' disease biomarkers.Alzheimers Res Ther. 2024 Jun 22;16(1):132. doi: 10.1186/s13195-024-01503-x. Alzheimers Res Ther. 2024. PMID: 38909218 Free PMC article.
-
Cerebrospinal fluid in Alzheimer's: A precious tool.Oncotarget. 2017 Jan 17;8(3):3770. doi: 10.18632/oncotarget.14157. Oncotarget. 2017. PMID: 28031540 Free PMC article. No abstract available.
-
Serum tau concentration after diving - an observational pilot study.Diving Hyperb Med. 2019 Jun 30;49(2):88-95. doi: 10.28920/dhm49.2.88-95. Diving Hyperb Med. 2019. PMID: 31177514 Free PMC article.
-
BACE1 Function and Inhibition: Implications of Intervention in the Amyloid Pathway of Alzheimer's Disease Pathology.Molecules. 2017 Oct 13;22(10):1723. doi: 10.3390/molecules22101723. Molecules. 2017. PMID: 29027981 Free PMC article. Review.
-
Inhibition of DREAM-ATF6 interaction delays onset of cognition deficit in a mouse model of Huntington's disease.Mol Brain. 2018 Mar 9;11(1):13. doi: 10.1186/s13041-018-0359-6. Mol Brain. 2018. PMID: 29523177 Free PMC article.
References
-
- Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR, Jr, Kaye J, Montine TJ, et al. Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement. 2011;7(3):280–92. doi: 10.1016/j.jalz.2011.03.003. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

