L-type calcium channels refine the neural population code of sound level
- PMID: 27605536
- PMCID: PMC5133292
- DOI: 10.1152/jn.00657.2016
L-type calcium channels refine the neural population code of sound level
Abstract
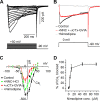
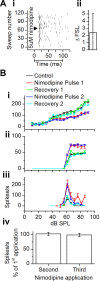
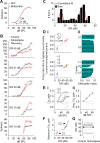
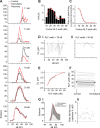
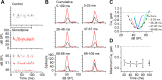
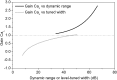
The coding of sound level by ensembles of neurons improves the accuracy with which listeners identify how loud a sound is. In the auditory system, the rate at which neurons fire in response to changes in sound level is shaped by local networks. Voltage-gated conductances alter local output by regulating neuronal firing, but their role in modulating responses to sound level is unclear. We tested the effects of L-type calcium channels (CaL: CaV1.1-1.4) on sound-level coding in the central nucleus of the inferior colliculus (ICC) in the auditory midbrain. We characterized the contribution of CaL to the total calcium current in brain slices and then examined its effects on rate-level functions (RLFs) in vivo using single-unit recordings in awake mice. CaL is a high-threshold current and comprises ∼50% of the total calcium current in ICC neurons. In vivo, CaL activates at sound levels that evoke high firing rates. In RLFs that increase monotonically with sound level, CaL boosts spike rates at high sound levels and increases the maximum firing rate achieved. In different populations of RLFs that change nonmonotonically with sound level, CaL either suppresses or enhances firing at sound levels that evoke maximum firing. CaL multiplies the gain of monotonic RLFs with dynamic range and divides the gain of nonmonotonic RLFs with the width of the RLF. These results suggest that a single broad class of calcium channels activates enhancing and suppressing local circuits to regulate the sensitivity of neuronal populations to sound level.
Keywords: auditory midbrain; dynamic range; inferior colliculus; level tuning; local circuits; rate-level functions.
Copyright © 2016 the American Physiological Society.
Figures


Similar articles
-
AMPA and NMDA receptors regulate responses of neurons in the rat's inferior colliculus.J Neurophysiol. 2001 Aug;86(2):871-80. doi: 10.1152/jn.2001.86.2.871. J Neurophysiol. 2001. PMID: 11495957
-
Differential regulation of action potential firing in adult murine thalamocortical neurons by Kv3.2, Kv1, and SK potassium and N-type calcium channels.J Physiol. 2007 Oct 15;584(Pt 2):565-82. doi: 10.1113/jphysiol.2007.141135. Epub 2007 Aug 30. J Physiol. 2007. PMID: 17761775 Free PMC article.
-
Dendritic voltage-gated ion channels regulate the action potential firing mode of hippocampal CA1 pyramidal neurons.J Neurophysiol. 1999 Oct;82(4):1895-901. doi: 10.1152/jn.1999.82.4.1895. J Neurophysiol. 1999. PMID: 10515978
-
Nimodipine inhibits AP firing in cultured hippocampal neurons predominantly due to block of voltage-dependent potassium channels.Gen Physiol Biophys. 2011;30 Spec No:S44-53. doi: 10.4149/gpb_2011_SI1_44. Gen Physiol Biophys. 2011. PMID: 21869451
-
Space-rate coding in an adaptive silicon neuron.Neural Netw. 2001 Jul-Sep;14(6-7):645-56. doi: 10.1016/s0893-6080(01)00082-x. Neural Netw. 2001. PMID: 11665760 Review.
Cited by
-
Tonotopic and non-auditory organization of the mouse dorsal inferior colliculus revealed by two-photon imaging.Elife. 2019 Oct 15;8:e49091. doi: 10.7554/eLife.49091. Elife. 2019. PMID: 31612853 Free PMC article.
-
Axonal and presynaptic FMRP: Localization, signal, and functional implications.Hear Res. 2023 Mar 15;430:108720. doi: 10.1016/j.heares.2023.108720. Epub 2023 Feb 11. Hear Res. 2023. PMID: 36809742 Free PMC article. Review.
References
-
- Bar PR, Traber J, Schuurman T, Gispen WH. CNS and PNS effects of nimodipine. J Neural Transm Suppl 31: 55–71, 1990. - PubMed
-
- Barone P, Clarey JC, Irons WA, Imig TJ. Cortical synthesis of azimuth-sensitive single-unit responses with nonmonotonic level tuning: a thalamocortical comparison in the cat. J Neurophysiol 75: 1206–1220, 1996. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

