Daclatasvir plus sofosbuvir, with or without ribavirin, achieved high sustained virological response rates in patients with HCV infection and advanced liver disease in a real-world cohort
- PMID: 27605539
- PMCID: PMC5099229
- DOI: 10.1136/gutjnl-2016-312444
Daclatasvir plus sofosbuvir, with or without ribavirin, achieved high sustained virological response rates in patients with HCV infection and advanced liver disease in a real-world cohort
Erratum in
-
Erratum: Daclatasvir plus sofosbuvir, with or without ribavirin, achieved high sustained virological response rates in patients with HCV infection and advanced liver disease in a real-world cohort.Gut. 2016 Dec;65(12):2060. doi: 10.1136/gutjnl-2016-312444corr1. Gut. 2016. PMID: 28157683 Free PMC article. No abstract available.
Abstract
Objective: We assessed the effectiveness and safety of daclatasvir (DCV) plus sofosbuvir (SOF), with or without ribavirin (RBV), in a large real-world cohort, including patients with advanced liver disease.
Design: Adults with chronic HCV infection at high risk of decompensation or death within 12 months and with no available treatment options were treated in a European compassionate use programme. The recommended regimen was DCV 60 mg plus SOF 400 mg for 24 weeks; RBV addition or shorter duration was allowed at physicians' discretion. The primary endpoint was sustained virological response at post-treatment week 12 (SVR12).
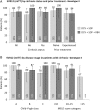
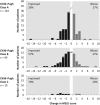
Results: Of the 485 evaluable patients, 359 received DCV+SOF and 126 DCV+SOF+RBV. Most patients were men (66%), white (93%) and treatment-experienced (70%). The most frequent HCV genotypes were 1b (36%), 1a (33%) and 3 (21%), and 80% of patients had cirrhosis (42% Child-Pugh B/C; 46% Model for End-Stage Liver Disease score >10). SVR12 (modified intention-to-treat) was achieved by 91% of patients (419/460); 1 patient had virological breakthrough and 13 patients relapsed. Virological failure was not associated with treatment group (adjusted risk difference DCV+SOF minus DCV+SOF+RBV: 1.06%; 95% CI -2.22% to 4.35%). High SVR12 was observed regardless of HCV genotype or cirrhosis, liver transplant or HIV/HCV coinfection status. Twenty eight patients discontinued treatment due to adverse events (n=18) or death (n=10) and 18 died during follow-up. Deaths and most safety events were associated with advanced liver disease and not considered treatment related.
Conclusions: DCV+SOF with or without RBV achieved high SVR12 and was well tolerated in a diverse cohort of patients with severe liver disease.
Trial registration number: NCT02097966.
Keywords: ANTIVIRAL THERAPY; CHRONIC VIRAL HEPATITIS; CIRRHOSIS; HEPATITIS C.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://www.bmj.com/company/products-services/rights-and-licensing/.
Conflict of interest statement
Figures


Comment in
-
Toward chronic hepatitis C eradication in HIV-positive patients, including those cirrhotic and infected with genotype 3 viruses.Gut. 2017 Oct;66(10):1868-1869. doi: 10.1136/gutjnl-2016-313431. Epub 2017 Jan 10. Gut. 2017. PMID: 28073889 No abstract available.
-
Mortality associated with the treatment of HCV with direct-acting antivirals.Gut. 2018 Jan;67(1):197-198. doi: 10.1136/gutjnl-2017-313869. Epub 2017 Mar 23. Gut. 2018. PMID: 28341750 No abstract available.
Similar articles
-
Daclatasvir plus sofosbuvir, with or without ribavirin, in real-world patients with HIV-HCV coinfection and advanced liver disease.Antivir Ther. 2017;22(3):225-236. doi: 10.3851/IMP3108. Epub 2016 Nov 15. Antivir Ther. 2017. PMID: 27845298
-
Real-world experience with daclatasvir plus sofosbuvir ± ribavirin for post-liver transplant HCV recurrence and severe liver disease.Transpl Int. 2017 Mar;30(3):243-255. doi: 10.1111/tri.12910. Transpl Int. 2017. PMID: 28012215
-
Efficacy and safety of sofosbuvir plus daclatasvir with or without ribavirin: large real-life results of patients with chronic hepatitis C genotype 4.Hepatol Int. 2018 Jul;12(4):348-355. doi: 10.1007/s12072-018-9868-8. Epub 2018 May 12. Hepatol Int. 2018. PMID: 29754329
-
Pharmacodynamic and pharmacokinetic evaluation of the combination of daclatasvir/sofosbuvir/ribavirin in the treatment of chronic hepatitis C.Expert Opin Drug Metab Toxicol. 2018 Sep;14(9):901-910. doi: 10.1080/17425255.2018.1506765. Epub 2018 Aug 2. Expert Opin Drug Metab Toxicol. 2018. PMID: 30058394 Review.
-
Daclatasvir and Sofosbuvir Versus Sofosbuvir and Ribavirin in Patients with Chronic Hepatitis C Coinfected with HIV: A Matching-adjusted Indirect Comparison.Clin Ther. 2016 Feb;38(2):404-12. doi: 10.1016/j.clinthera.2015.12.017. Epub 2016 Feb 3. Clin Ther. 2016. PMID: 26839044
Cited by
-
2017 KASL clinical practice guidelines management of hepatitis C: Treatment of chronic hepatitis C.Clin Mol Hepatol. 2018 Sep;24(3):169-229. doi: 10.3350/cmh.2018.1004. Epub 2018 Aug 10. Clin Mol Hepatol. 2018. PMID: 30092624 Free PMC article. Review. No abstract available.
-
Consensus on management of hepatitis C virus infection in resource-limited Ukraine and Commonwealth of Independent States regions.World J Gastroenterol. 2019 Aug 7;25(29):3897-3919. doi: 10.3748/wjg.v25.i29.3897. World J Gastroenterol. 2019. PMID: 31413526 Free PMC article. Review.
-
Hepatitis C Guidance 2018 Update: AASLD-IDSA Recommendations for Testing, Managing, and Treating Hepatitis C Virus Infection.Clin Infect Dis. 2018 Oct 30;67(10):1477-1492. doi: 10.1093/cid/ciy585. Clin Infect Dis. 2018. PMID: 30215672 Free PMC article.
-
Treatment of HCV infection with direct-acting antiviral agents. Real life experiences from the Euro-Asian region.Turk J Gastroenterol. 2020 Feb;31(2):148-155. doi: 10.5152/tjg.2020.19440. Turk J Gastroenterol. 2020. PMID: 32141824 Free PMC article.
-
Rapid platelet count improvement in chronic hepatitis C patients with thrombocytopenia receiving direct-acting antiviral agents.Medicine (Baltimore). 2020 May;99(19):e20156. doi: 10.1097/MD.0000000000020156. Medicine (Baltimore). 2020. PMID: 32384505 Free PMC article.
References
-
- American Association for the Study of Liver Diseases and the Infectious Diseases Society of America. Recommendations for testing, managing, and treating hepatitis C. http://www.Hcvguidelines.org/full-Report-View (accessed 21 Apr 2015).
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
